2.3. Basic energy science
2.3.2. Photosynthesis
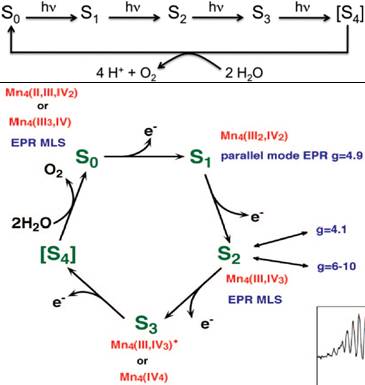
Oxygen is vital for all aerobic life forms and is abundant in the atmosphere due to continuous regeneration by photosynthetic water oxidation in green plants, algae, and cyanobacteria. This photosynthetic reaction is catalyzed by a Mn4CaO5 cluster located inside the photosystem II membrane protein complex. This cluster undergoes four consecutive redox states known as the Si (i = 1 to 4) states by the consecutive absorption of four photons by the PS II reaction center (Kok cycle). The accumulated energy is used for water oxidation and the catlyst returns to the S0 state in the Kok cycle (Figure 23).
Figure 23. Reaction cycle for water oxidation in PSII with the four states S0-S4 reached by consecutive absorption of four photons [top, taken from J. Kern et al., Science, 14 February 2013 DOI: 10.1126/science.1234273 (2013)] and with the corresponding proposed oxidation states of the Mn cluster [bottom, adapted from ref. 1, J. Yano, V. Yachandra, Inorg. Chem. 47, 1711 (2008)].
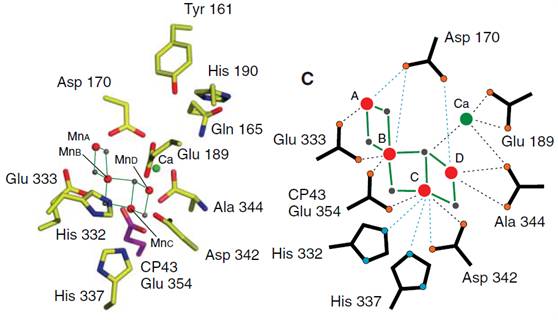
Figure 24. Two representations of a model representing the immediate vicinity of the Mn4CaO5 cluster in PSII. The assignment of ligands is tentative because it is based on the electron density of the cluster, and its immediate environment may be altered by x-ray damage. Bonds in the representation on the right between Mn and the bridging oxo are shown as solid green lines. Bonds to proposed ligand amino acid groups and to Ca atoms are shown as dotted lines (black, less than 3.0 Å; blue, more than 3.0 Å). Taken from ref. 2, J. Yano et al., Science 314, 824 (2006).
Understanding the mechanism of the photosynthetic water oxidation on a molecular level can be considered to be one of sciences grand challenges (Figure 24). The structure and catalytic activity of PSII have been studied intensively, but a description of the photochemical processes on a molecular level all the way from photon absorption to water-oxidation is still largely missing. Explaining the reaction kinetics though requires knowledge of the dynamics pathways of the Mn4CaO5 cluster.
This is essential to answer several key questions for understanding the mechanism of photosynthetic water oxidation (Figure 24). Is Mn(V) involved during O-O bond formation and, if yes, in which spin state? How does the ligand environment modulate the Mn d orbitals and the electronic structure of the Mn cluster? How does this relate to the change of the redox potential during the catalytic process? Are water molecules bound to the cluster and do they participate in the oxidation? What is the role of the Ca atom in the cluster? How does it bind to the ligands and to the Mn atoms?
BESSY-VSR with its few-ps pulses at MHz repetition rate could uniquely complement a series of investigations recently started at the Linac Coherent Light Source (LCLS, USA) using diffraction to reveal the geometric structure of the catalytic center and femtosecond spectroscopy to address the electronic structure of the Mn4CaO5 cluster. The pulse length at BESSY-VSR of a few picoseconds will be important in two respects: First, it will be essential to overcome X-ray damage to the catalytic center reported from studies at conventional synchrotrons with pulse lengths in the 100 ps domain [3]. At BESSY-VSR we can anticipate to be able to probe the system before the X-ray radiation destroys it. For this, the protein will be studied at ambient conditions and ideally in solution at a millimolar concentration. Second, BESSY-VSR with its high repetition rate compared to LCLS (MHZ compared to Hz) and accordingly with its better signal to noise ratio will enable studying more subtle changes of the electronic structure accompanying the catalytic activity on a nanosecond and millisecond time scale.
Time-resolved Mn and Ca L-edge X-ray absorption spectroscopy could be used to map the electronic structure locally around the Mn and Ca atoms in the Mn4CaO5 cluster in photosystem II. An optical laser would be used to advance the complex throughout the catalytic cycle. By varying the delay between the visible-pump and X-ray-probe pulses dynamics of the water-oxidation reaction will be revealed on a pico- to millisecond time scale.
References for section 2.3.2.
[1] J. Yano, V. Yachandra, Inorg. Chem. 47, 1711 (2008).
[2] J. Yano et al., Science 314, 824 (2006).
[3] J. Yano et al., PNAS 102, 12047 (2005).