IRIS beamline at BESSY II extended with nanomicroscopy
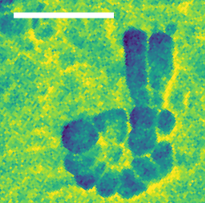
Infrared image of the nucleolus in the nucleus of a fibroblast cell. The scale bar corresponds to 500 nanometres. © HZB
The IRIS infrared beamline at the BESSY II storage ring now offers a fourth option for characterising materials, cells and even molecules on different length scales. The team has extended the IRIS beamline with an end station for nanospectroscopy and nanoimaging that enables spatial resolutions down to below 30 nanometres. The instrument is also available to external user groups.
The infrared beamline IRIS at the BESSY II storage ring is the only infrared beamline in Germany that is also available to external user groups and is therefore in great demand. Dr Ulrich Schade, in charge of the beamline, and his team continue to develop the instruments to enable unique, state-of-the-art experimental techniques in IR spectroscopy.
As part of a recent major upgrade to the beamline, the team, together with the Institute of Chemistry at Humboldt University Berlin, has built an additional infrared near-field microscope.
"With the nanoscope, we can resolve structures smaller than a thousandth of the diameter of a human hair and thus reach the innermost structures of biological systems, catalysts, polymers and quantum materials," says Dr Alexander Veber, who led this extension.
The new nanospectroscopy end station is based on a scanning optical microscope and enables imaging and spectroscopy with infrared light with a spatial resolution of more than 30 nm. To demonstrate the performance of the new end station, Veber analysed individual cellulose microfibrils and imaged cell structures. All end stations are available to national and international user groups.
Funding information: Bundesministerium für Bildung und Forschung [grant No. project 05K19KH1 (SyMS)]; Germany's Excellence Strategy (grant No. EXC 2008-390540038 – UniSysCat).
arö
https://www.helmholtz-berlin.de/pubbin/news_seite?nid=26746;sprache=en
- Copy link
-
AI agents deliver results but do they reason scientifically?
A research team co-led by Kevin Maik Jablonka from the Helmholtz Institute for Polymers in Energy Applications Jena (HIPOLE Jena) and N. M. Anoop Krishnan from the Indian Institute of Technology Delhi has developed Corral, a new benchmark for AI agents in science. The preprint “AI scientists produce results without reasoning scientifically” has been published on arXiv (https://doi.org/10.48550/arXiv.2604.18805). The analysis shows that current systems can execute scientific workflows and deliver results; however, they often do not follow the basic principles of scientific testing and reasoning.
-
Magnetic field during catalyst synthesis triples ammonia yield
Applying an external magnetic field during the synthesis of CoFe₂O₄ electrocatalysts triples the ammonia yield during electrocatalytic conversion. The magnetic field alters the surface states of the spinel oxide thin films, making catalytically active sites more accessible. In the journal 'Advanced Functional Materials', a team led by Marcel Risch at HZB and Sanjay Mathur at University of Cologne demonstrates a scalable strategy for developing next-generation electrocatalysts for efficient and sustainable chemical production.
-
Materials chemistry shapes the future of catalysis
The synthesis of materials can serve as a tool for developing smart, adaptive electrocatalysts. This rapidly evolving field of research involves in-situ analytics, data-driven discoveries and autonomous robotics. These new approaches could accelerate the discovery of long-lasting and efficient catalysts for future energy conversion and the decarbonisation of the chemical industry. A recent article by Dr Prashanth Menezes and his team in the renowned journal Angewandte Chemie provides an overview of this research.
