EMIL
Catalysis
Nearly 50% of the world’s supply of bioavailable ammonia results from the industrial Haber-Bosch process. The annual ammonia production is 200 Mt. It consumes 1-2 % of the world energy supply, and it generates about 400 Mt of greenhouse gases annually. However, the detailed mechanism of how the triple bond of dinitrogen is activated by iron surfaces in industrial catalysis has remained elusive and many open questions remain regarding the nature of formed surface nitrides, the oxidation state of the active iron and the role of promoters.
Industrial ammonia synthesis
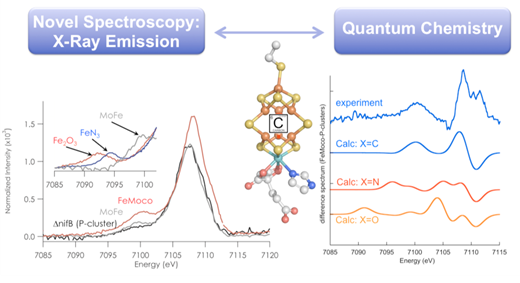
Much of our current understanding is based on pioneering studies by Ertl, Somoraji and many others, who investigated idealized iron surfaces. Ertl and coworkers proposed Fe2N nitrides formed at the surface and that a metastable Fe4N formed in situ, while Herzog and coworkers proposed the formation of subnitrides with a Fe(10-20)N composition. However, to date the formation of iron-nitride phases in the ammonia synthesis catalysts have yet to be directly observed. Some argue that this is due to the limited range of experimental methods that may be readily applied to the high temperature and high-pressure operating conditions (200 bar and 500 C) of the industrial catalyst. In this context, hard x-ray probes may provide an ideal means to obtain direct insight into the detailed mechanism.
Biological ammonia synthesis
The reduction of dinitrogen to bioavailable ammonia is a process fundamental for life. In biology, this is enabled by the nitrogenase enzymes, which contain complex Fe7S9M metalloclusters at the active site (where M=Mo, V or Fe). The reactivity of the enzymes varies greatly, depending on the identity of M. However, the site of substrate interaction remains unknown. Does the heterometal (M) serve as a substrate binding site? Or does it simply tune the electronic structure for optimal reactivity? The answers to these questions are key to understanding how the biological active site is able to break the triple bond of nitrogen under ambient temperatures and pressures.