User research at BESSY II: Graphite electrodes for rechargeable batteries investigated
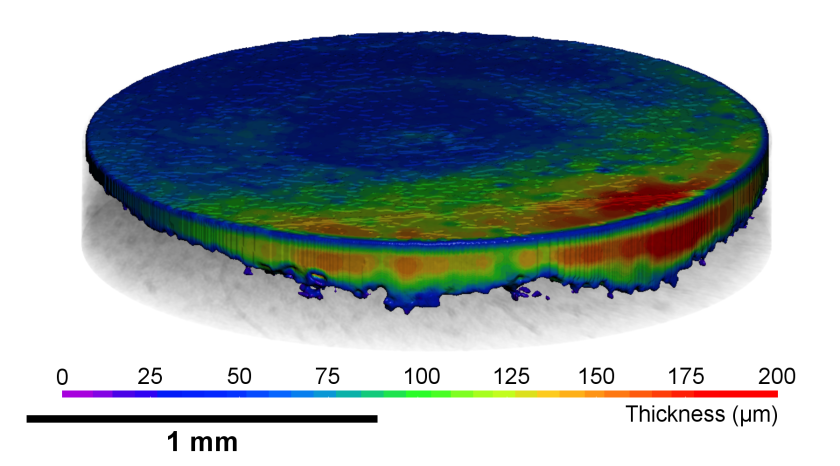
The tomogram during the charging process shows the spatially resolved changes in the graphite electrode thickness of a rechargeable aluminium ion battery in a discharged and charged state. © HZB
Rechargeable graphite dual ion batteries are inexpensive and powerful. A team of the Technical University of Berlin has investigated at the EDDI Beamline of BESSY II how the morphology of the graphite electrodes changes reversibly during cycling (operando). The 3D X-ray tomography images combined with simultaneous diffraction now allow a precise evaluation of the processes, especially of changes in the volume of the electrodes. This can help to further optimise graphite electrodes.
Published in Advanced Functional Materials (2020); Simultaneous X‐Ray Diffraction and Tomography Operando Investigation of Aluminum/Graphite Batteries; Giuseppe Antonio Elia, Giorgia Greco, Paul Hans Kamm, Francisco García‐Moreno, Simone Raoux, Robert Hahn
DOI: 10.1002/adfm.202003913
Abstract: Rechargeable graphite dual‐ion batteries are extremely appealing for grid‐level stationary storage of electricity, thanks to the low‐cost and high‐performance metrics, such as high‐power density, energy efficiency, long cycling life, and good energy density. An in‐depth understanding of the anion intercalation mechanism in graphite is fundamental for the design of highly efficient systems. In this work, a comparison is presented between pyrolytic (PG) and natural (NG) graphite as positive electrode materials in rechargeable aluminum batteries, employing an ionic liquid electrolyte. The two systems are characterized by operando synchrotron energy‐dispersive X‐ray diffraction and time‐resolved computed tomography simultaneously, establishing a powerful characterization methodology, which can also be applied more in general to carbon‐based energy‐related materials. A more in‐depth insight into the AlCl4−/graphite intercalation mechanism is obtained, evidencing a mixed‐staged region in the initial phase and a two‐staged region in the second phase. Moreover, strain analysis suggests a correlation between the irreversibility of the PG electrode and the increase of the inhomogenous strain. Finally, the imaging analysis reveals the influence of graphite morphology in the electrode volume expansion upon cycling.
red.
https://www.helmholtz-berlin.de/pubbin/news_seite?nid=22334;sprache=en
- Copy link
-
Spintronics at BESSY II: Real-time analysis of magnetic bilayer systems
Spintronic devices enable data processing with significantly lower energy consumption. They are based on the interaction between ferromagnetic and antiferromagnetic layers. Now, a team from Freie Universität Berlin, HZB and Uppsala University has succeeded in tracking, for each layer separately, how the magnetic order changes after a short laser pulse has excited the system. They were also able to identify the main cause of the loss of antiferromagnetic order in the oxide layer: the excitation is transported from the hot electrons in the ferromagnetic metal to the spins in the antiferromagnet.
-
Electrocatalysts: New model for charge separation at the solid-liquid interface
Hydrogen is at the heart of the transition to carbon neutrality, as both an energy carrier and a reagent for green chemistry. However, large-scale production of hydrogen via electrolysis, as well as the production of many other chemical products, requires significantly cheaper and more efficient catalysts. A precise understanding of the electrochemical processes that take place at the interface between the solid catalyst and the liquid medium is highly useful for developing better electrocatalysts. In the journal Nature Communications, an European team has now presented a powerful model that determines charge separation at the interface, the formation of the electric double layer and local electric potential variations, and the resulting influence on the catalytic activity.
-
Environmental Chemistry at BESSY II: Radicals in waterways
How do radicals form in aqueous solutions when exposed to UV light? This question is important for health research and environmental protection, for example with regard to the overfertilisation of water bodies by intensive agriculture. A team at BESSY II has now developed a new method of investigating hydroxyl radicals in solution. By using a clever trick, the scientists gained surprising insights into the reaction pathway.
