Young Investigator Group Electrochemical Conversion
Sustainable synthesis through electrocatalysis
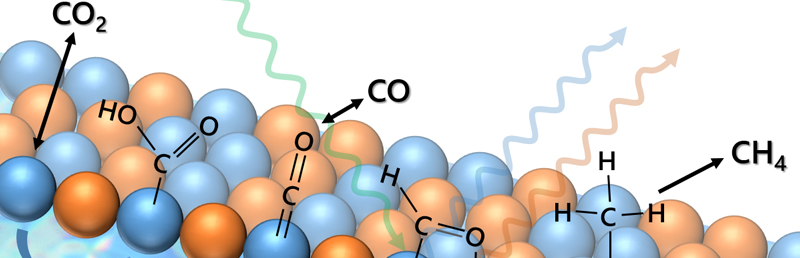
Multi-metal electrocatalysis - Controlling the mechanistic pathways for electrochemical CO2 reduction
The Electrochemical Conversion of CO2 group studies how carbon dioxide and water can be converted electrochemically into valuable products such as hydrocarbons and alcohols, using renewable energy as driving force.
Presently, most carbon-based fuels and industrial chemicals originate from fossil resources accumulated over millions of years. Following their use, these fuels and chemicals decompose to CO2 which is released into our atmosphere, disturbing the carbon equilibrium and inducing changes in the global climate.Research Topics
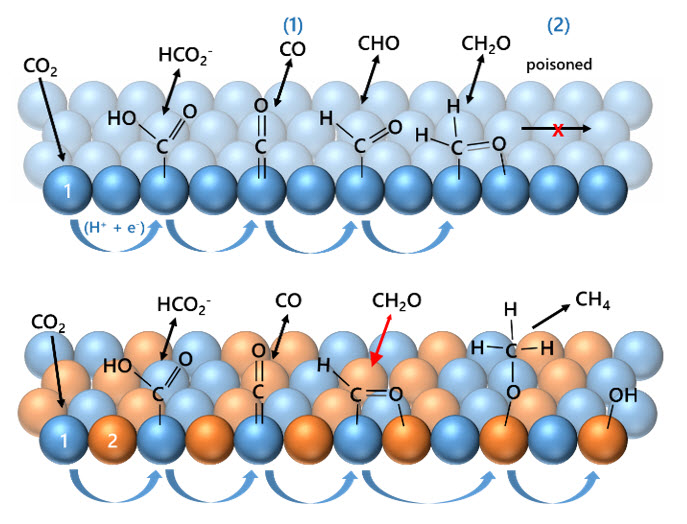
Structureactivity relationships in bi-metallic catalysts
Our goal is to electrochemically convert CO2 to highly-reduced, valuable organic products (alkanes, alkenes, alcohols) in a single process. Individual metal catalysts suffer from binding energy scaling, which impart thermodynamic barriers and result in low efficiency and poor yield of the desired products. We will examine the ability of multi-metal catalysts to enable different binding modes and reaction mechanisms which favor the production of highly-reduced products.
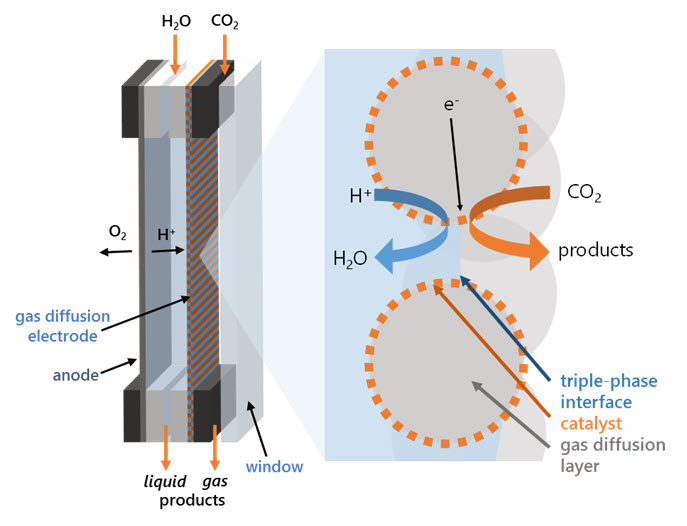
Targeting kinetic limitations via advanced cell design
The low solubility of CO2 in water limits conventional systems to low current densities. For the technology to be industrially viable, much higher rates of CO2 transport are necessary, and this can be achieved by using gas diffusion cells. Our research efforts involve synthesis of robust membrane–electrode assemblies suited for electrochemical CO2 reduction, and design of cells capable of efficient product collection.
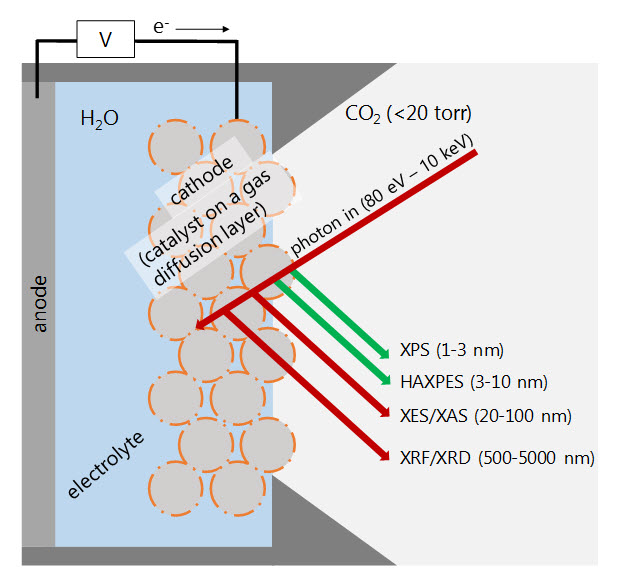
Mechanism study using operando spectroscopy
Improved understanding of electrochemical mechanisms is needed to enable design of better catalysts. We are developing methods for spectroscopic analysis of catalysts under real operation conditions. This includes design of an electrochemical cell for X-ray emission/absorption and photoelectron spectroscopy.
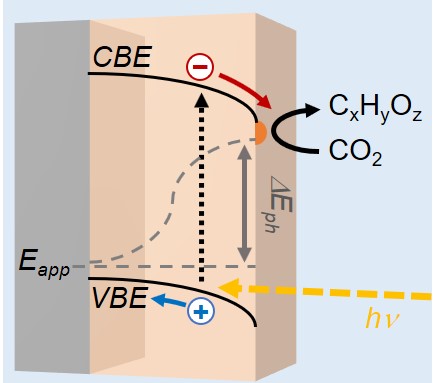
Photoelectrochemical phenomena
Semiconductor electrodes give different activity than metallic electrodes. Due to their electronic band structure, the energy distribution of charges is more discrete than for metals, with a strong dependence on the electrode material and the semiconductor-electrolyte interface. Furthermore, they can be activated by light, enabling photo-to-chemical energy conversion, and additional photoelectrochemical phenomena which we aim to exploit for new control over reaction selectivity.