BESSY II: New insights into switchable MOF structures at the MX beamlines
View into a MOF crystal exemplified by DUT-8. The massive pores are clearly discernible. © TU Dresden
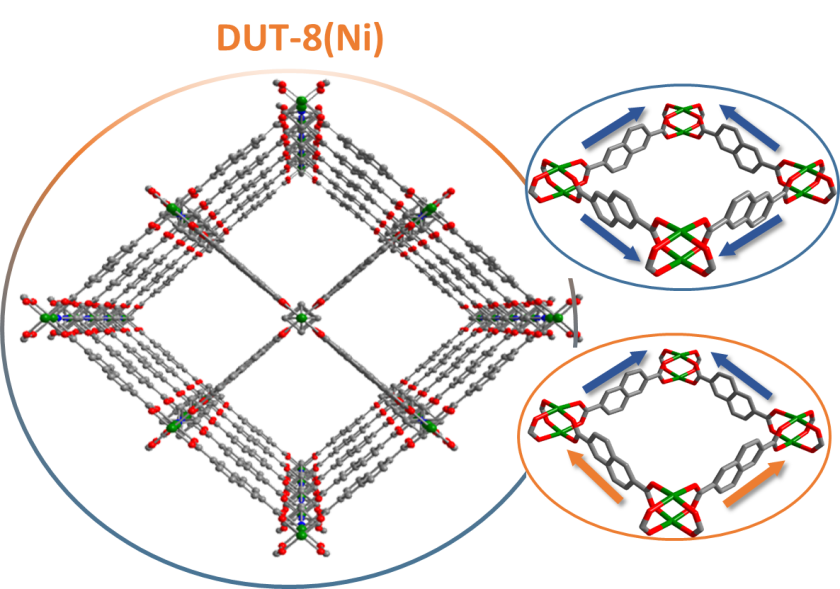
Metal-organic framework compounds (MOFs) are widely used in gas storage, material separation, sensor technology or catalysis. A team led by Prof. Dr. Stefan Kaskel, TU Dresden, has now investigated a special class of these MOFs at the MX beamlines of BESSY II. These are "switchable" MOFs that can react to external stimuli. Their analysis shows how the behaviour of the material is related to transitions between ordered and disordered phases. The results have now been published in Nature Chemistry.
Metal-organic framework compounds (MOFs) consist of inorganic and organic groups and are characterised by a large number of pores into which other molecules can be incorporated. MOFs are therefore interesting for many applications, for example for the storage of gases, but also for substance separation, sensor technology or catalysis. Some of these MOF structures react to different guest molecules by changing their structures. They are thus considered switchable.
One of these is "DUT-8", a material that has now been studied at the MX beamlines of BESSY II. "MOF crystals can be analysed very well at the MX beamlines," says HZB expert Dr. Manfred Weiss, who heads the MX team. “MOF crystals have many things in common with protein crystals. For example, both are interspersed with large pores, which are filled with liquid in the protein crystals, while those in MOFs provide space for guest molecules," Weiss explains.
"The diffraction patterns that DUT-8 showed on the HZB-MX beamlines were extremely complex. We were now able to attribute this to various transitions between ordered and less ordered phases," explains Stefan Kaskel. The enclosed guest molecule directs the network into one of over a thousand possible disorder configurations. The results contribute to a better understanding of switching processes and gas exchange reactions in such MOF structures, so that future functional MOF materials can be developed in a targeted manner.
The investigations were supported by the DFG programme (FOR2433).
red.
https://www.helmholtz-berlin.de/pubbin/news_seite?nid=22861;sprache=en
- Copy link
-
Materials chemistry shapes the future of catalysis
The synthesis of materials can serve as a tool for developing smart, adaptive electrocatalysts. This rapidly evolving field of research involves in-situ analytics, data-driven discoveries and autonomous robotics. These new approaches could accelerate the discovery of long-lasting and efficient catalysts for future energy conversion and the decarbonisation of the chemical industry. A recent article by Dr Prashanth Menezes and his team in the renowned journal Angewandte Chemie provides an overview of this research.
-
Imaging Ellipsometry for Process Control of Thin-Film Devices
A German–Israeli research team led by Dr. Andreas Furchner has demonstrated how imaging ellipsometry enables non-destructive characterisation and quality control of microstructured MXene thin films during device fabrication. The authors used two complementary ellipsometry approaches for precise, multi-scale access to key material properties. The work positions imaging ellipsometry as a powerful platform for monitoring thin-film uniformity, device integrity, and functionality throughout processing, including critical lithographic steps. The study was published in Applied Physics Letters and selected as an Editor’s Pick.
-
Cool vaccines in rural Kenya: solar solution has been awarded by UN
In May 2026, Tabitha Awuor Amollo is spending some weeks as a guest scientist at HZB, analysing perovskite thin films at BESSY II. The Kenyan physicist from Egerton University, Nairobi, was recently recognised for her achievements in research and teaching. For the development of a solar-powered refrigeration system for use in rural health centres, she has been awarded the 2026 Organization for Women in Science for the Developing World (OWSD)-Elsevier Foundation Award. An interview on exceptional projects and daily struggles of a scientist. Questions were asked by Antonia Rötger.
