Göttingen scientists use BESSY II to decode basic mechanism underlying biochemical reactions
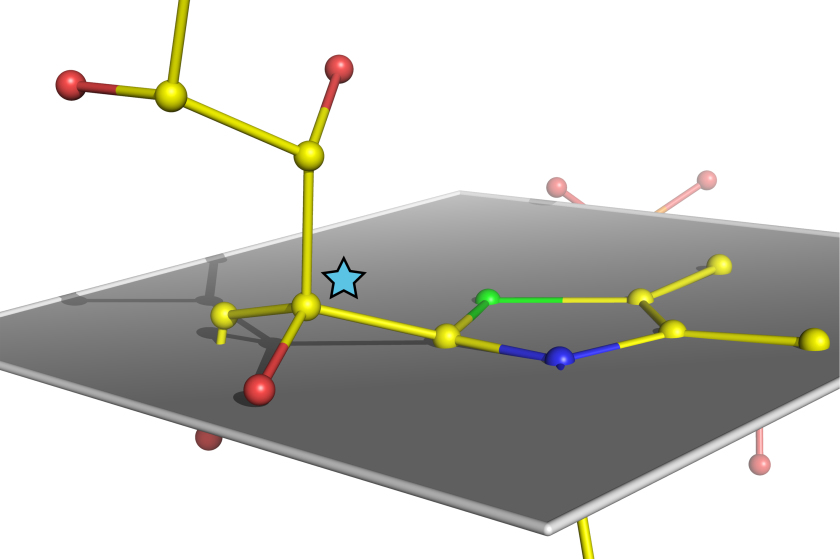
Structure of the sugar molecule bound by the enzyme transketolase immediately prior to its being split
Enzymes are life’s molecular catalysts and figure prominently in cellular metabolism. It has been speculated that in the course of a biochemical reaction enzymes physically bend their substrates to split them. Now for the first time ever, scientists at the Göttingen Center for Molecular Biosciences (GZMB) have successfully used BESSY II's MX beamline to unequivocally confirm this hypothesis. The results from this study have been published in the renowned scientific journal Nature Chemistry.
The Göttingen team around Prof. Dr. Kai Tittmann and Prof. Dr. Ralf Ficner started out by growing high order protein crystals of the human enzyme transketolase, which plays a central role in human metabolism during sugar processing. Natural sugar substrates were added to the protein crystals. Analysis of the enzyme’s crystalline structure was subsequently performed at electron storage ring BESSY II's MX beamline and in French Grenoble. The scientists were able to determine the structure of the sugar molecule bound by the enzyme immediately prior to its being split in half at an ultrahigh spatial resolution of 0.1 nanometers. “The snapshot we got of an enzyme at work, which really is unprecedented in terms of resolution, unequivocally reveals how the sugar substrate is being bent by the enzyme, similar to a vise clamping a work piece,” Prof. Tittmann explains.
In many cases, enzymes are drug targets. Which is why these new insights are important for the development of customized, highly specific active substances like those used in cancer therapy. “Even the human transketolase used in this study plays a key role in cancer cell metabolism,” says Prof. Tittmann.
Source: Göttingen University
https://www.helmholtz-berlin.de/pubbin/news_seite?nid=13800;sprache=en
- Copy link
-
BESSY II: How intrinsic oxygen shortens the lifespan of solid-state batteries
Although solid-state batteries (SSBs) demonstrate high performance and are intrinsically safe, their capacity currently declines rapidly. A team from the TU Wien, Humboldt-University Berlin and HZB has now analysed a TiS₂|Li₃YCl₆ solid-state half-cell in operando at BESSY II using a special sample environment that allows for non-destructive investigation under real operating conditions. Data obtained by combination of soft and hard X-ray photoelectron spectroscopy (XPS and HAXPES) revealed a new degradation mechanism that had not previously been identified in solid-state batteries. They have gained some surprising insights, particularly regarding the harmful role played by intrinsic oxygen. This study provides valuable information for improving design and handling of such batteries.
-
Spintronics at BESSY II: Real-time analysis of magnetic bilayer systems
Spintronic devices enable data processing with significantly lower energy consumption. They are based on the interaction between ferromagnetic and antiferromagnetic layers. Now, a team from Freie Universität Berlin, HZB and Uppsala University has succeeded in tracking, for each layer separately, how the magnetic order changes after a short laser pulse has excited the system. They were also able to identify the main cause of the loss of antiferromagnetic order in the oxide layer: the excitation is transported from the hot electrons in the ferromagnetic metal to the spins in the antiferromagnet.
-
Environmental Chemistry at BESSY II: Radicals in waterways
How do radicals form in aqueous solutions when exposed to UV light? This question is important for health research and environmental protection, for example with regard to the overfertilisation of water bodies by intensive agriculture. A team at BESSY II has now developed a new method of investigating hydroxyl radicals in solution. By using a clever trick, the scientists gained surprising insights into the reaction pathway.
