Institute Solar Fuels
Currently available Student Projects
We can offer a variety of projects in our institute to students from universities in Berlin and beyond.
Should you be interested in pursuing a bachelor’s or master’s thesis with us, please choose one of our BSc/MSC projects (listed below), and send your application with your CV and a list of the courses that you have followed, directly to the responsible project leader.
Insights into PEC device scaling limitations at near-neutral pH: In-situ measurement of voltage losses caused by electrolyte pH gradients
Target group:
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Students of chemical engineering, chemistry or a related field. Strong understanding of electrochemistry and mass transport in liquids. Prior experience in Multiphysics simulations or hands-on experience in (photo-) electrochemistry is a plus.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
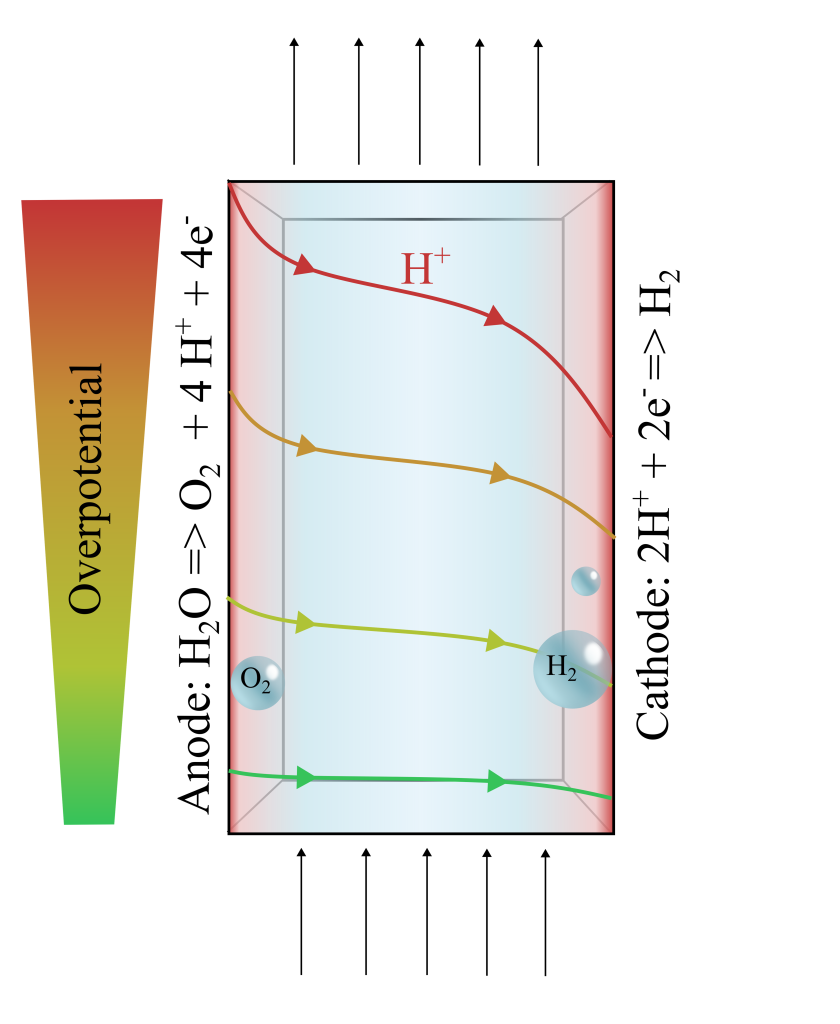
Project description:
Photo-electrochemical (PEC) reactors are a promising prospect for green hydrogen production or conversion of hydrocarbons into valuable chemicals directly from sunlight. The key to the success of PEC is maximizing device efficiency and enhancing the photoelectrode stability, which are difficult to achieve both at the same time. Near-neutral electrolytes offer a less harsh environment with significantly enhanced device stability. However, during water splitting they show severe overpotentials due to mass transport limitations caused by their low proton concentration. While buffered electrolytes under a steady flow can improve the supply of protons between the electrodes, there may be a limitation of the buffer at larger electrode lengths.
In this project you will explore a novel method to directly measure the increasing overpotential, supported by multi-physics simulations.
You will:
- Perform Comsol Multiphysics simulations to predict voltage losses under different operationparameters.
- Help to set up a novel technique to directly measure pH-gradient overpotentials over 10 cm long electrodes.
- Replicate the simulations experimentally and correlate the results.
- Conclude operation restrictions for PEC modules.
Optimizing the fluid dynamics in photoelectrochemical reactors
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Students of chemical engineering, chemistry or a related field. Strong understanding of electrochemistry and mass transport in liquids. Prior experience in Multiphysics simulations or hands-on experience in (photo-) electrochemistry is a plus.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
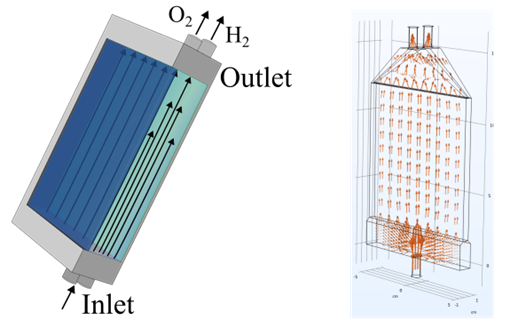
Project description:
Photo-electrochemical reactors are a promising prospect for green hydrogen production or conversion of hydrocarbons into valuable chemicals directly from sunlight. As a conjunction of solar cell, and electrolyzer, their complexity is rooted in the requirement to efficiently balance the transport of light and charge in the photoabsorber, while maintaining efficient mass transport for the electrochemistry. While past research of small-scale devices mostly focused on the light and charge transport in the heart of the device, the photoelectrode, the hydrodynamic optimization for the transport of reactants and products in the liquid electrolyte have been often overlooked and remain a key engineering limitation for large scale reactors. Efficient device inlet diffusors and reactor outlet shapes are required to ensure uniform flow distribution over the photoelectrode, steady product removal without dead zones, while minimizing the size of flow guides and their pressure loss in the overall device plant.
The project aims to find optimized inlet and outlet designs for a 10 x 10 cm2 reactor module for water splitting and/or glycerol oxidation. The project will be divided into two parts, with emphasis on the students’ interest:
- CAD design and computational fluid dynamic (CFD) simulations in Comsol Multiphysics to predetermine promising inlet diffusor types and outlet geometries
- 3D printing a prototype and confirming simulations using particle image velocimetry, dye-tests, and/or bubble shadowgraphy under (photo-)electrochemical operation.
Investigation of pulsed electrolysis and flow-engineering for enhanced selectivity in photoelectrochemical (PEC) glycerol oxidation
Target group:
- BSc thesis (6 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
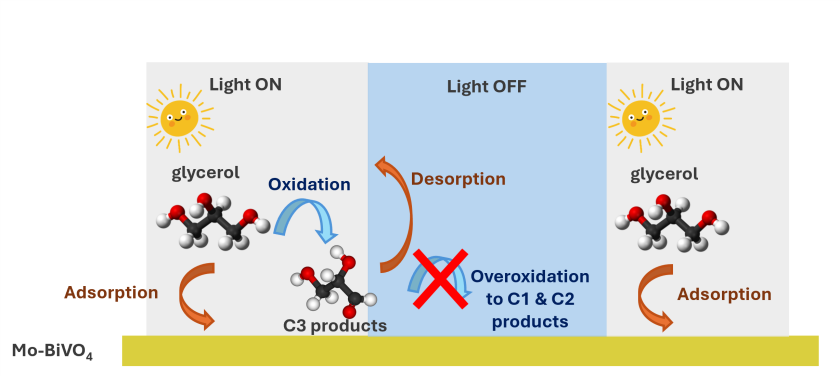
Project description:
This project aims to investigate the influence of dynamic operating conditions on product selectivity
in the photoelectrochemical glycerol oxidation reaction (GOR). Pulsed electrolysis has recently emerged as a promising strategy to modulate reaction pathways and improve selectivity in organic electrosynthesis, offering temporal control over interfacial reaction environments.1,2
The primary objective is to determine whether the distribution of oxidation products is governed
by external operating parameters, specifically variations in flow regime, applied potential, and light irradiation profiles.
The experimental work will involve the preparation of molybdenum-doped bismuth vanadate (Mo-BiVO₄) photoanodes via a well-established electrodeposition method. These photoanodes will be characterised in both conventional static PEC cells and flow-cell configurations to assess the role of mass transport.
Dynamic operation will be implemented through controlled voltage and light pulsing using a range
of waveform profiles (e.g. square, sinusoidal). The resulting photoelectrochemical performance and product distribution under glycerol oxidation conditions will be systematically evaluated. Quantitative analysis of reaction products will be conducted using high-performance liquid chromatography (HPLC).
The findings are expected to contribute to the rational design of selective and energy-efficient processes for biomass valorisation, in alignment with the objectives of the PH2OTOGEN project.
- Atkins, A. P. & Lennox, A. J. J. Application of pulsed electrolysis in organic electrosynthesis. Curr. Opin. Electrochem. 44, 101441 (2024)
- Chen, W. et al. Pulse potential mediated selectivity for the electrocatalytic oxidation of glycerol to glyceric acid. Nat. Commun. 15, 1–11 (2024)
Successive Ionic Layer Adsorption and Reaction (SILAR) of tungsten-doped BiVO₄ (W-BVO) thin films and decoration with a co-catalyst for photoelectrochemical (PEC) selective glycerol oxidation reaction (GOR)
Target group:
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee

Project description:
This project aims to develop efficient and selective photoanodes for the PEC GOR through controlled fabrication of W-BVO thin films and their functionalization with β-Bi₂O₃ co-catalysts.
The first part of the study focuses on optimizing the SILAR process1 for the reproducible deposition
of homogeneous W-BVO films on conductive substrates, including FTO and porous Ti felt. In addition,
the introduction of a WO₃ underlayer will be investigated to enhance charge separation and improve PEC performance.
The second part addresses the development of a selective co-catalyst via electrodeposition of β-Bi₂O₃ nanoparticles. Emphasis will be placed on controlling nanoparticle morphology and surface coverage
to maximize selectivity towards value-added products. Enhancing selectivity remains a central challenge in GOR, with previous studies indicating that β-Bi₂O₃-modified systems can achieve up to ~75% selectivity towards dihydroxyacetone (DHA).2
The photoelectrochemical performance and product distribution will be evaluated under glycerol oxidation conditions, with quantitative analysis performed using high-performance liquid chromatography (HPLC).
Overall, this work seeks to establish a robust synthesis strategy for W-BVO photoanodes with improved activity and selectivity, contributing to the development of sustainable routes for biomass valorization within the framework of the PH2OTOGEN project.
- Zafeiropoulos, G. et al. Rational Design of Photoelectrodes for the Fully Integrated Polymer Electrode Membrane-Photoelectrochemical Water-Splitting System: A Case Study of Bismuth Vanadate. ACS Appl. Energy Mater. 4, 9600–9610 (2021)
- Luo, L. et al. Selective Photoelectrocatalytic Glycerol Oxidation to Dihydroxyacetone via Enhanced Middle Hydroxyl Adsorption over a Bi2O3-Incorporated Catalyst. J. Am. Chem. Soc. 144, 7720–7730 (2022)
Development of an optimized HPLC Method for product analysis in the glycerol oxidation reaction (GOR) in photoelectrochemical (PEC) cell
Target group:
- BSc thesis (6 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
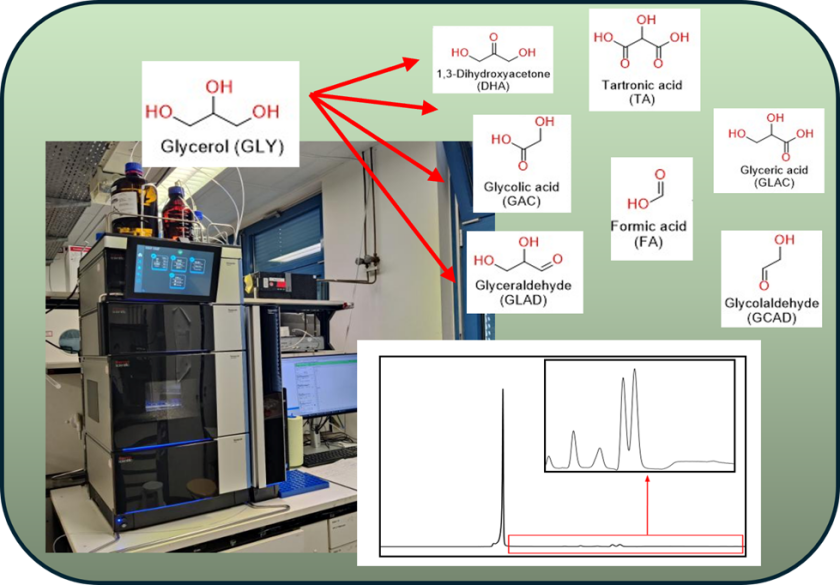
Project description:
The glycerol oxidation reaction (GOR) has been extensively investigated as a promising anodic alternative to the oxygen evolution reaction (OER) in electrochemical and photoelectrochemical (PEC) systems. Compared to OER, GOR operates at lower potentials and enables the generation of value-added chemicals, thereby improving both the energy efficiency and economic feasibility of electrochemical processes. A variety of oxidation products can be obtained from glycerol, including dihydroxyacetone (DHA), glyceraldehyde (GLAD), glyceric acid (GLAC), glycolic acid (GAC), tartronic acid (TA), formic acid (FA), and oxalic acid (OXAC). These compounds differ significantly in their chemical properties and market value. Consequently, accurate identification and quantification of the product distribution are essential for evaluating the reaction performance.
High-performance liquid chromatography (HPLC) is a suitable analytical technique for this purpose. However, due to the structural similarity and polarity of the products, efficient peak separation and reliable detection remain challenging. Therefore, a systematic optimization of HPLC parameters is required to establish a robust analytical method.
The primary objective of this thesis is to develop and optimize an HPLC-based analytical method for the separation, identification, and quantification of GOR products. Specific goals include:
- Systematic investigation of HPLC operating parameters, including:
- Column temperature
- Eluent composition and flow rate
- Detector type and settings
- Development of an efficient and reproducible method for peak separation and compound identification.
- Application of the developed method to analyze product distributions from GOR experiments in a PEC cell.
Investigation of lignin oxidation reaction in photoelectrochemical (PEC) cell employing BiVO4
Target group:
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
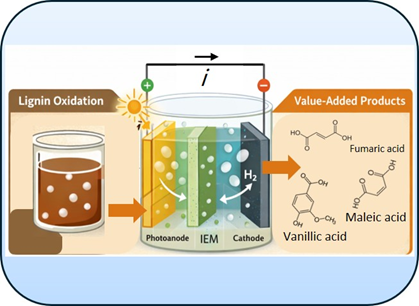
Project description:
In recent years, there has been increasing interest in replacing the oxygen evolution reaction (OER) in photoelectrochemical (PEC) cells with alternative reactions to improve efficiency and economic viability. In this context, biomass valorization is particularly attractive, as it enables operation at lower potentials while simultaneously generating value-added chemicals. Among the investigated substrates, glycerol has received considerable attention; however, its global annual production is limited to approximately 7 million tons. In contrast, lignin, an abundant byproduct of the pulp and paper industry with an annual production exceeding 70 million tons, represents a more scalable feedstock and its oxidation can yield high-value compounds such as vanillic acid, which are relevant for various industrial applications.
Therefore, the primary objective of this thesis is to investigate the photoelectrochemical oxidation of lignin in a membrane-based PEC cell. Specific goals include evaluating photoelectrochemical performance, identifying key reaction parameters that influence activity, and optimizing reaction conditions.
The following activities will be a part of this Master thesis:
- Preparation and characterization of the doped and undoped BiVO4 photoanodes
- Investigation of the optimal electrolyte composition (solvent; conductive salt; pH; concentration;..) for the lignin oxidation in a PEC cell on prepared BiVO4 photoanode
- Assessment of photoanode stability and degradation under operating conditions
Understand the growth of cobalt on alumina thin film
Target group:
- BSc thesis (6 months)
Language:
English
Prerequisite:
Knowledge in inorganic chemistry and physical chemistry.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
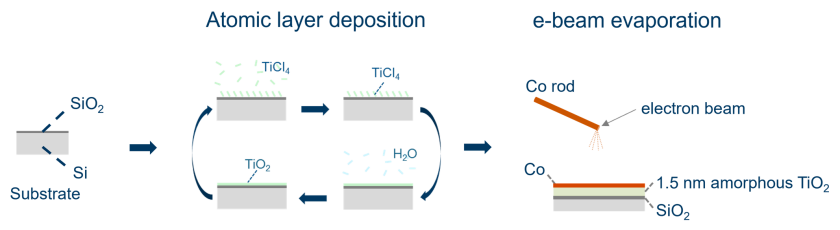
Project description:
Fischer-Tropsch catalysis is a key catalytic process for producing sustainable aviation fuels from alternative carbon sources. It is inherently complex due to the wide distribution of long-chain hydrocarbon products and the structural complexity of industrial catalysts, which are typically optimized for both activity and selectivity. This complexity makes it difficult to isolate and understand fundamental phenomena such as metal-support interactions, which play a crucial role in catalyst performance and stability.
Metal-support interactions can significantly influence cobalt-based catalysts. On one hand, strong interactions may enhance cobalt dispersion, leading to a higher density of active sites and improved catalytic activity. On the other hand, they may also induce partial encapsulation of cobalt nanoparticles by the support, thereby blocking active sites and contributing to catalyst deactivation. A systematic understanding of these effects is essential for developing strategies to design more active and stable catalysts.
To address this challenge, model catalyst systems provide a simplified and well-controlled platform. In particular, thin film-based catalysts allow precise structural control and are well suited for surface-sensitive characterization techniques under in situ conditions, which are often difficult to apply to conventional powder catalysts. Atomic layer deposition (ALD) enables the fabrication of uniform and well-defined oxide support films such as alumina, while electron beam evaporation allows controlled deposition of cobalt with sub-monolayer precision.
This project aims to investigate the growth behaviour of cobalt on alumina thin films as a representative of insulator support. By combining ALD-grown oxide films with cobalt deposition via electron beam evaporation, the study will focus on how cobalt nucleates and evolves on the surface. X-ray photoelectron spectroscopy (XPS) will be employed to analyse the chemical state and growth mode of cobalt. Understanding these fundamental growth mechanisms is a critical step toward designing well-defined model catalysts for future in situ studies of metal–support interactions.
Understanding the growth of titania thin film via atomic layer deposition using tetrakis(dimethylamino)titanium
Target group:
- BSc thesis (6 months)
Language:
English
Prerequisite:
Knowledge in inorganic chemistry and physical chemistry.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
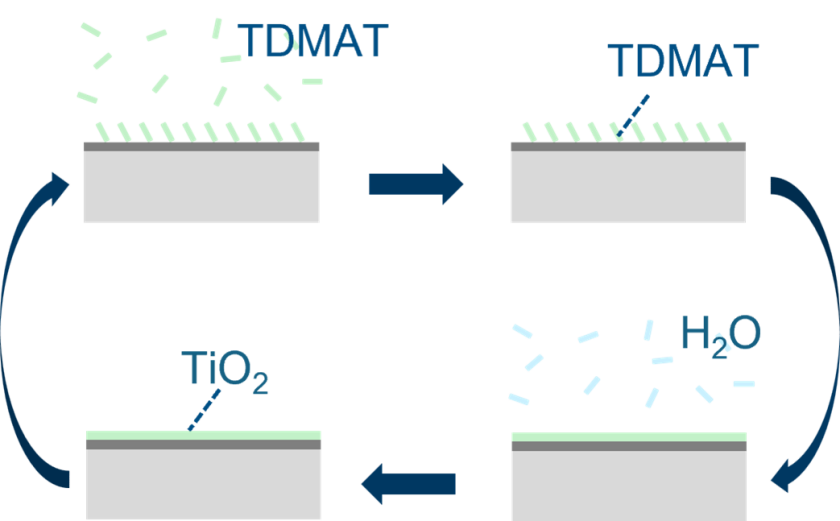
Project description:
Model catalysts provide a powerful approach for systematically studying surface interactions under well-defined conditions. Such systems usually rely on single-crystal substrates, which offer precise control over atomic sites. However, these simplified systems do not capture metal–support interactions, a key factor in determining the performance of industrial catalysts. In practical applications, catalysts typically consist of metal nanoparticles dispersed on oxide supports, where strong metal–support interactions can significantly influence catalytic activity, selectivity, and stability.
In the context of Fischer-Tropsch synthesis, the interaction between cobalt and titania is particularly important for tuning catalytic performance. To bridge the gap between idealized model systems and real catalysts, this project focuses on developing a model catalyst system based on oxide thin films. Specifically, titania thin films will be prepared as supports using atomic layer deposition (ALD), a technique that enables precise structural control at the nanometer scale. Importantly, these thin films remain sufficiently conductive to allow characterization with surface-sensitive techniques.
Titania films can be deposited using different precursors, such as titanium tetrachloride (TiCl₄) or tetrakis(dimethylamino)titanium (TDMAT). In this project, TDMAT is chosen due to its halogen-free and non-corrosive nature, offering a cleaner and potentially more controllable deposition route.
The main objective is to understand the growth behaviour of TiO₂ on passivated silicon wafers and to demonstrate structural control over the resulting thin films. The student will carry out ALD experiments to synthesize the films and employ X-ray photoelectron spectroscopy (XPS) to investigate the film growth behaviours.
Modeling an X-ray Absorption & Photoelectron Spectroscopy (XAS & XPS) Experiment at BElChem-DCM using Python
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Basic programming knowledge
Location:
HZB Wilhelm-Conrad-Röntgen-Campus
Albert-Einstein-Straße 15
12489 Berlin-Adlershof

A simplified workflow for modeling an XAS & XPS experiment at the BElChem-DCM beamline using python.
Project description:
This project will provide a detailed understanding of XAS and XPS experiments, by numerically tracking the synchrotron radiation and X-ray optics as well as the core-level spectroscopy techniques. An XAS and/or XPS experiment will be designed and carried out using one of our standard samples: (1) BiVO4 thin film, (2) [Cu(dmp)2](PF6) molecules in solution, or (3) Lanthanide-based nanoparticles in solution. The optimal sample preparation/delivery method will be discussed and chosen to address scientific questions for the sample. The X-ray absorption cross-section and its corresponding fluorescence/electron yield will be estimated by taking advantage of existing python libraries. The resulting XAS and/or XPS spectra will be compared to the experimental data, eventually answering our scientific questions.
Background:
The Berlin Joint Lab for Electrochemical Interfaces (BElChem) is located at the BESSY II synchrotron in Berlin-Adlershof and consists of two beamlines – one is BElChem-PGM (plane-grating monochromator), and the other is BElChem-DCM (double-crystal monochromator). The latter receives the synchrotron radiation from a dipole bending magnet at the BESSY II. The X-ray is tailored by 2 cylindrical mirrors and 1 pair of Si(111) crystals, i.e., DCM, and focused to <300 µm in full width at half maximum (FWHM). The BElChem-DCM is operating in the photon energy range between 2.4 and 10 keV and equipped with the Spectroscopic Analysis with Tender X-rays (SpAnTeX) end-station. This end-station can accommodate a sample environment for studying solid/liquid, solid/gas, and liquid/gas interfaces and reactions. X-ray absorption spectrum can be measured in both transmission and fluorescence modes. X-ray photoemission spectrum can be measured using the hemispherical electron analyser designed for ambient pressure X-ray photoelectron spectroscopy (AP-XPS).
Spectroscopic Investigation of Nanoscale Systems for Solar Energy Conversion
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Basic analytical chemistry
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
&
HZB Wilhelm-Conrad-Röntgen-Campus
Albert-Einstein-Straße 15
12489 Berlin-Adlershof
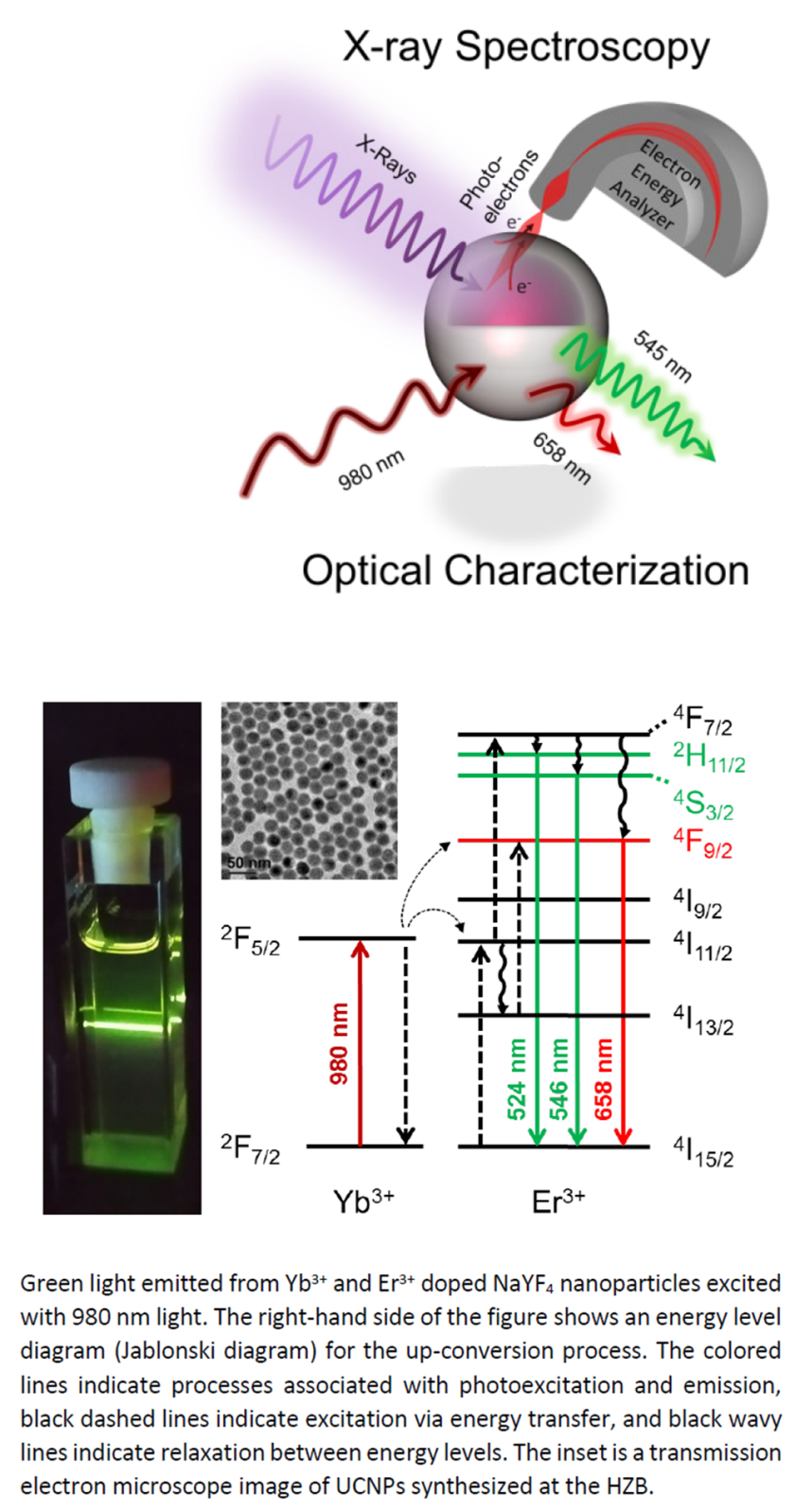
A simplified workflow for modeling an XAS & XPS experiment at the BElChem-DCM beamline using python.
Project description:
This project will introduce an energy transfer process called photon up-conversion. Inorganic materials with ions of d-block or f-block elements often show photon up-conversion phenomena, e.g., lanthanide ion (Ln3+)-based nanoparticles and molecular clusters absorb infrared and emit visible light. The goal of the project is to understand their energy migration pathways and quenching mechanism in the excited states by means of optical and X-ray spectroscopies. This will involve (1) synthesis, (2) characterization of Ln3+-based nanoparticles and/or molecular clusters, and eventually (3) theoretical modelling of the observed signals and kinetics. These will enable a strategic design of new upconverting materials and a deeper understanding of interatomic cross-over dynamics upon photoabsorption.
Background:
Ln3+-based crystalline nanoparticles and hetero-nonnuclear clusters are representative systems exhibiting up-conversion luminescences (infrared à visible). This process typically involves Yb3+ ions as a sensitizer to absorb ~980 nm light and Er3+, Tb3+, or Tm3+ ions as an activator to emit photons from higher excited states. The emitted wavelength however differs by the activator element – for example, Er3+ emits green/red light whereas Tm3+ emits blue light. A reproducible, high-yield synthesis of Ln3+-based systems has enabled their applications in solar energy conversion and bioimaging. The detailed energy transfer and relaxation pathways are yet to be studied, which are essential for achieving higher quantum yield and wavelength tunability. Our unique approach to studying these materials is a newly constructed droplet train experiment coupled to X-ray spectroscopies at the BESSY II synchrotron. The time structure of the droplet train allows time-resolved measurements on the micro- to milli-second timescales, which is ideally matched to the excited state lifetimes of Ln3+-based upconverting materials. We use synchrotron-based ambient pressure X-ray photoelectron spectroscopy (AP-XPS) and X-ray absorption spectroscopy (XAS) as well as lab-based optical techniques to study these systems.
Plasmon-enhanced electrochemical CO2 reduction to C2+ product
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
A simplified workflow for modeling an XAS & XPS experiment at the BElChem-DCM beamline using python.
Project description:
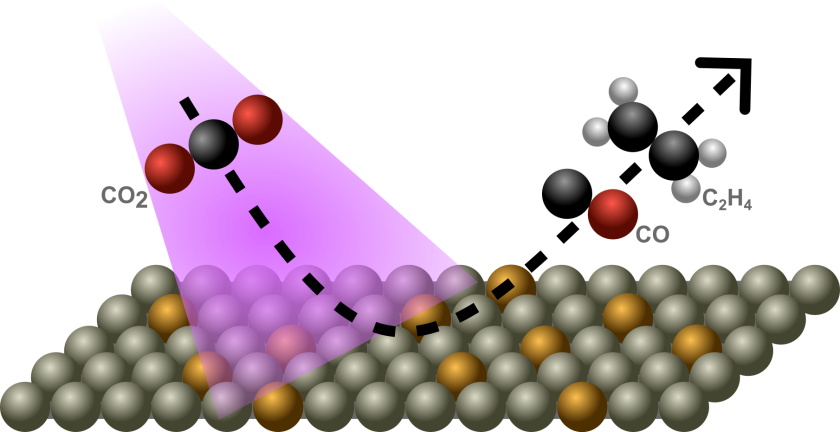
Electrochemical CO2 reduction (CO2RR) is a promising strategy to convert the greenhouse gas CO2 into valuable chemicals and fuels. However, when CO2 reduction is carried out in aqueous electrolytes, the reaction often competes with the hydrogen evolution reaction (HER), which reduces the efficiency and selectivity of CO2 conversion. One potential strategy to steer reaction selectivity is to use plasmonic excitation. Plasmonic metals such as Au, Ag, and Cu can generate hot electrons when illuminated at their surface plasmon resonance (SPR) frequency. These hot electrons may participate in catalytic reactions by transferring to adsorbed molecules. In the case of CO2 reduction, the rate-determining step is often the activation of CO2 via electron transfer to form the radical anion CO2•-. In principle, hot electrons generated under plasmonic excitation could preferentially promote this activation step and thereby enhance CO2 reduction over HER. Previous studies have shown that plasmon illumination of Au or Ag catalysts can indeed increase the selectivity of CO2RR towards CO. However, Au and Ag primarily produce C1 products (CO) and cannot efficiently generate more complex multi-carbon products. In contrast, Cu-based catalysts are unique in their ability to form C2+ products (e.g. ethylene and ethanol), although they typically produce a mixture of products with limited selectivity. The aim of this project is therefore to investigate whether plasmonic excitation can be used to influence product selectivity in Cu-based CO2 reduction systems towards C2+ products, which is a relatively unexplored area.
The experimental work will be divided into two phases:
Phase 1:
In the first phase, the student will reproduce and study plasmon-assisted CO2 reduction using well-established Au or Ag systems. Metal nanoparticles will be deposited on gas diffusion electrodes (GDEs) to ensure efficient CO2 mass transport. Photoelectrochemical experiments will be performed under plasmonic illumination, and gaseous products will be monitored online using mass spectrometry. This phase will establish the experimental setup and verify the plasmonic enhancement of CO2RR over HER.
Phase 2:
In the second phase, Cu will be incorporated into the catalytic system in order to explore plasmon-assisted formation of C2+ products. Different strategies for integrating Cu with plasmonic structures will be explored, for example Cu as standalone plasmonic catalysts or Cu catalysts coupled with plasmonic nanoparticles. The goal is to investigate whether plasmon-generated hot carriers can influence reaction pathways and improve the selectivity towards C2+ products.
Effect of particle size and distribution on plasmon-enhanced electrochemical CO2 reduction
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
A simplified workflow for modeling an XAS & XPS experiment at the BElChem-DCM beamline using python.
Project description:
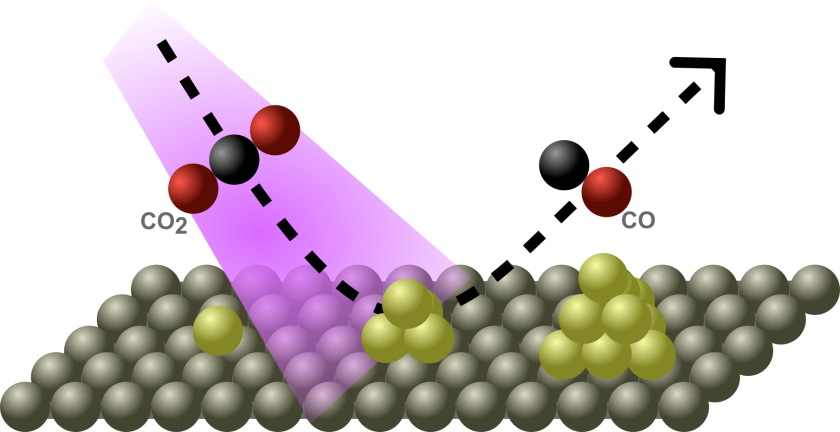
Electrochemical CO2 reduction (CO2RR) is a promising strategy to convert the greenhouse gas CO2 into valuable chemicals and fuels. However, when CO2 reduction is carried out in aqueous electrolytes, the reaction often competes with the hydrogen evolution reaction (HER), which reduces the efficiency and selectivity of CO2 conversion. One potential strategy to steer reaction selectivity is to use plasmonic excitation. Plasmonic metals such as Au, Ag, and Cu can generate hot electrons when illuminated at their surface plasmon resonance (SPR) frequency. These hot electrons may participate in catalytic reactions by transferring to adsorbed molecules. In the case of CO2 reduction, the rate-determining step is often the activation of CO2 via electron transfer to form the radical anion CO2•-. In principle, hot electrons generated under plasmonic excitation could preferentially promote this activation step and thereby enhance CO2 reduction over HER. However, the efficiency of hot carrier generation strongly depends on the size and spatial distribution of the plasmonic nanoparticles. Small nanoparticles tend to favor optical absorption over scattering, which is beneficial for hot carrier generation. In contrast, larger particles or particle agglomerates typically exhibit stronger scattering contributions, which can reduce the efficiency of hot electron generation. These considerations suggest that small, well-dispersed nanoparticles may be optimal for plasmon-enhanced catalysis. Despite this expectation, a systematic investigation of how nanoparticle size and aggregation affect plasmon-assisted CO2 reduction remains largely unexplored.
The experimental work will be divided into two phases:
Phase 1:
In the first phase, Ag nanoparticles with different sizes will be synthesized and deposited onto gas diffusion electrodes (GDEs) to enable efficient CO2 mass transport. Photoelectrochemical measurements will then be performed to evaluate how nanoparticle size influences the plasmon-enhanced activity and selectivity for CO2 reduction.
Phase 2:
In the second phase, the same Ag nanoparticles will be deposited using different preparation methods to control their degree of dispersion or agglomeration on the electrode surface. By comparing well-dispersed particles with intentionally agglomerated structures, the project will investigate how nanoparticle distribution affects plasmonic enhancement in CO2 reduction.
Mechanism of glycerol oxidation on BiVO4
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
Project description:
Glycerol -an abundant by-product of the biodiesel industry- is often treated as waste. But what if it could be transformed into high-value chemicals used in pharmaceuticals, cosmetics, and food?
Photo)electrochemical glycerol oxidation offers exactly this opportunity. However, there is a major challenge: selectivity. The reaction can generate more than 11 different products, but only a few are economically valuable. Controlling which products are formed, and under what conditions, is key to making this process viable.
In this project, you will tackle this challenge by investigating the reaction mechanism of glycerol oxidation using BiVO₄ as a model photoanode. Instead of focusing only on performance, the goal is to understand how and when different products are formed.
Using a custom-designed Zahner cell (large electrode area, small electrolyte volume), you will be able to rapidly accumulate reaction products and quantify them using HPLC. By systematically oxidizing glycerol and its intermediates at increasing overpotentials -guided by their thermodynamic oxidation potentials- you will map out the reaction network and identify the conditions under which side products emerge.
The experimental work will be divided into two main phases:
Phase 1:
Photoelectrochemical oxidation of glycerol and key intermediates on BiVO₄. The student will perform controlled electrochemical measurements at different applied potentials and concentrations, to probe activity and product formation.
Phase 2:
Product analysis and reaction pathway mapping. Using HPLC, the student will quantify products and correlate their formation with applied potential, identifying thresholds where selectivity is lost and side reactions become dominant.
This project will provide fundamental insight into potential-dependent reaction pathways, helping to answer a central question in electrochemical biomass upgrading: How can we control selectivity in complex reaction networks?
Multi metal co-doping of BiVO4 for glycerol upgrading
Target group:
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
Project description:
Can sunlight be used not only to generate energy - but also to produce valuable chemicals?
In conventional photoelectrochemical systems, the oxygen evolution reaction (OER) limits efficiency due to its slow kinetics. A promising alternative is the oxidation of biomass-derived molecules such as glycerol, which is both thermodynamically more favourable and capable of producing high-value chemicals. However, achieving high activity, stability, and selectivity remains a major challenge.
Bismuth vanadate (BiVO₄) is a leading photoanode material for solar-driven oxidation reactions, but it suffers from poor charge transport and recombination losses. Doping strategies have shown significant improvements in performance by enhancing conductivity and modifying defect chemistry. More recently, co-doping has emerged as a powerful approach to simultaneously tune bulk electronic properties and surface catalytic behaviour.
In this project, the student will investigate how co-doping BiVO₄ with multiple metal cations affects its performance in glycerol photoelectrooxidation. Special attention will be given not only to photocurrent and stability, but also to how dopants influence reaction pathways and product selectivity.
The experimental work will be divided into two main phases:
Phase 1:
Synthesis and characterization of pristine and co-doped BiVO₄ photoanodes. The student will prepare materials with different dopant combinations and investigate their structural, optical, and electronic properties using techniques such as XRD, SEM, UV-Vis-spectroscopy, and electrochemical methods (e.g., voltammetry, chronoamperometry and impedance spectroscopy).
Phase 2:
Photoelectrochemical testing and glycerol oxidation studies. The student will evaluate photocurrent response and operational stability under illumination, followed by product analysis using chromatographic techniques (HPLC) to determine selectivity toward value-added compounds.
Buffer effect on the photoelectrochemical stability and selectivity of Gd and La-doped BiVO4 photoanodes towards glycerol valorization
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Knowledge in chemistry. Special interest in electrochemistry, materials science and environmental chemistry is also important. Experience with HPLC is not required, but it would be considered a plus.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
Project description:
The use of BiVO4 photoanodes is a known strategy in the field of photoelectrochemistry to promote sustainable low-carbon hydrogen production by the water-splitting process. However, BiVO4 suffers from poor stability and fast charge recombination, which implies the development of new strategies to improve its performance.
This thesis project will focus on different strategies to increase the performance of BiVO4 photoanodes: (I) structure modification by doping with gadolinium and lanthanum, both rare earth elements; and (II) replacing the conventional water splitting process by the oxidation of glycerol (GLY) in different buffers to modulate the GLY selectivity by changing the media.
The experimental activity will be divided into the following phases:
Phase 1:
Synthesis of La and Gd-doped BiVO4 thin films by electrodeposition. In this phase, the student will prepare different doped samples with different content of lanthanides (0.25%, 0.5%, and 1%) and investigate the effect of the lanthanide insertion in the morphology and physical properties of the obtained materials.
Phase 2:
Effect of the La and Gd insertion into the photoelectrochemical activity of the prepared materials. In this phase, the student will perform voltammetry investigations in different buffer compositions (sulfate pH 2, phosphate pH 7 and carbonate pH 10) to verify the prepared material activity towards water splitting and GLY oxidation processes.
Phase 3:
Effect of the buffer composition into the selectivity of GLY oxidation using the doped samples. The most promising materials studied in phase 2 will be tested in long-time photo-electrolysis to verify the material stability and selectivity to generate different GLY oxidation products. In this phase, the development of analytical calibration curves using high-performance liquid chromatography with ultraviolet and refraction index detectors (HPLC/UV-RID) will be also done to identify the generated products.
Comparing the insertion of rare earth elements into BiVO4 photoanodes by different electrochemical synthesis
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Knowledge in chemistry. Special interest in electrochemistry, materials science and environmental chemistry is also important. Experience with materials physical characterization is not required, but it would be considered a plus.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
Project description:
The use of BiVO4 photoanodes is a known strategy in the field of photoelectrochemistry to promote sustainable low-carbon hydrogen production by the water-splitting process. However, BiVO4 suffers from poor stability and fast charge recombination, which implies the development of new strategies to improve its performance.
This thesis project will focus on exploring different strategies to obtain La and Gd doped BiVO4 photoanodes doped with 1% of each lanthanide. The student will work to obtain new materials and test the influence of the lanthanides (Ln) insertion into the physical and photoelectrochemical properties. For the BiVO4 obtention, a two-step synthesis will be employed: (I) electrochemical deposition of Bi or BiOI, and (II) thermal conversion after the addition of V precursor by dropwise into the Bi or BiOI surface. The student will investigate the effect of the Ln insertion during steps I or II and also compare the effect of changing the first step synthesis between Bi and BiOI.
The experimental activity will be divided into the following phases:
Phase 1:
Ln insertion by electrochemical co-deposition. During this phase, the student will obtain the doped materials by adding the Ln into the electrochemical bath to obtain Ln-BiOI films before thermal conversion. The prepared materials will be physical and photoelectrochemical characterized by different techniques such as scanning electron microscopy (SEM), X-ray diffraction (XRD), Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), and voltammetry.
Phase 2:
Ln insertion by dropwise addition. During this phase, the student will obtain the doped materials by adding the Ln together with V during the synthesis step II, after the BiOI film formation. The materials will be characterized by the same techniques described in “Phase 1”. In this phase, a comparison between Phase 1 and Phase 2 will be performed to understand the role of changing the step of Ln insertion into the prepared materials activity.
Phase 3:
Comparing Bi and BiOI synthesis. Under the optimized conditions observed into “Phase 2”, the student will investigate the effect of change the first step electrodeposition BiOI obtention by Bi.
Phase 4:
Degradation of contaminants by using the materials. After the optimization of the synthesis, the student will have the opportunity to apply the materials to investigate the photoelectrocatalytic degradation of a dye or an antibiotic as a real application of the new material for environmental remediation.
Enhancing transparent current collectors in photoelectrode systems for solar fuel production
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Students of chemical engineering, chemistry or physical engineering with interest in solar fuels, photoelectrochemistry and scaling up of electrochemical processes. With experience in electrochemistry, solid-state semiconductors and/or electrodeposition techniques.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee

Project description:
The production of solar fuels and value-added chemicals using exclusively solar energy has emerged as a compelling alternative to traditional fossil-fuel-based energy conversion. While promising, photoelectrochemical (PEC) processes must overcome significant engineering constraints before industrial-scale deployment is feasible. This requires transitioning from cm2-scale laboratory devices to the m2-scale systems necessary for outdoor operation.
Photoelectrode systems, where the conversion of energy to chemicals occurs, typically consist of a transparent substrate, a current collector, a photoabsorber, and a catalyst. For PEC processes to be viable, these photoelectrodes must be produced cost-effectively at scale, featuring high-efficiency components while minimising resistive losses. However, most synthesis methods are optimized for electrodes of approx. 1 cm2. When scaled to areas exceeding 100 cm2, these systems often suffer from high ohmic losses, film inhomogeneities, and premature degradation. Fluorine-doped tin oxide (FTO) is the standard transparent current collector for metal oxide photoelectrodes. However, its typical sheet resistance (7 to 14 Ω/sq) is inadequate for large-scale applications. To mitigate this, current collectors can be engineered to improve their effective resistivity, hence minimising ohmic losses during operation and preventing inhomogeneities during film synthesis.
This project aims to identify and optimize the most effective strategy for producing large-scale (>100 cm2) transparent current collectors for BiVO4 photoanodes. Two primary strategies will be compared: (i) sputtering of FTO grids [1] and (ii) electrodeposition of metallic (e.g. gold or nickel) grids [2]. The resulting current collectors will be utilized for the electrodeposition of BiVO4 thin-films [3]. Finally, these will be evaluated in a scaled-up PEC reactor for solar water splitting or glycerol oxidation.
- J. Capitão, et al., Journal of Power Sources 654 (2025) 237788
- I.Y. Ahmet, et al., Advanced Materials Interfaces 9 (2022) 220161
- H. Kong, et al., Chem. Sci. 15 (2024) 10425–10435
Modelling two-phase flow in photoelectrochemical reactors: the impact of bubble-size distribution on device performance
Target group:
- BSc thesis (6 months)
- MSc thesis (6-12 months)
Language:
English
Prerequisite:
Students of chemical engineering, chemistry or physical engineering with interest in solar fuels, photoelectrochemistry and scaling up of electrochemical processes. With experience in electrochemistry, electrolysers, computational fluid mechanics and/or Comsol Multiphysics.
Location:
HZB Lise-Meitner-Campus
Hahn-Meitner-Platz 1
14109 Berlin-Wannsee
Project description:
The synthesis of solar fuels represents a transformative shift away from fossil-fuel-dependent energy conversion. While photoelectrochemical (PEC) reactors offer a promising pathway, they have yet to reach industrial-scale deployment due to several unresolved engineering challenges. Key among these are the efficient evolution of gaseous products on photoelectrode surfaces and the subsequent downstream separation of these gases.
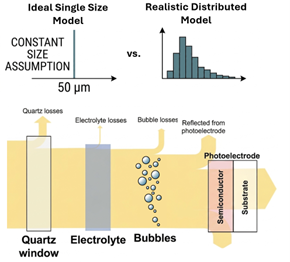
Traditional electrolysers operate at high current densities, typically between 0.1 and 2 A cm-2. Consequently, existing models for fluid mechanics and bubble evolution are optimized for this current density range. In contrast, PEC reactors typically operate at current densities between 1 and 10 mA cm-2, nearly three orders of magnitude lower. Under these conditions, two-phase (gas-liquid) flow dynamics behave differently, characterized by laminar flow regimes, lower operating temperatures and pressures, and much lower gas yields. While various models exist for predicting bubbly flow in PEC reactors [1], they often rely on the simplifying assumption of a constant bubble size (ca. 50 µm). In practice, bubble-size distribution varies both spatially and temporally. The ramifications of varying bubble sizes on PEC performance have been seldom explored, despite their significant impact on phenomena such as gas crossover in membrane-less devices.
This project aims to assess the effect of bubble-size distribution on PEC reactor performance by decoupling optical and ohmic losses. A two-phase (Euler-Euler) Computational Fluid Dynamics (CFD) model will be developed and enhanced using a Size-Based Population Balance Model (PBM) in Comsol Multiphysics. This approach will account for non-Gaussian bubble-size distributions on a platinized electrode surface for hydrogen evolution reaction. The model aims to predict gas evolution, hydrogen yields and collection efficiencies under moderate, laminar flow regimes (Re < 2000) at atmospheric pressure.
The project will focus on a specific case study: hydrogen evolution at the photocathode coupled with glycerol oxidation at the photoanode. Performance metrics will account for localized ohmic resistance and light attenuation caused by bubble coverage. Ultimately, this model will inform the design and loss budget estimation for scaled-up PEC reactors, facilitating the efficient production of solar fuels and value-added chemicals.
- F. E. Bedoya-Lora, et al., Electrochimica Acta 462 (2023) 14270
