New detector accelerates protein crystallography
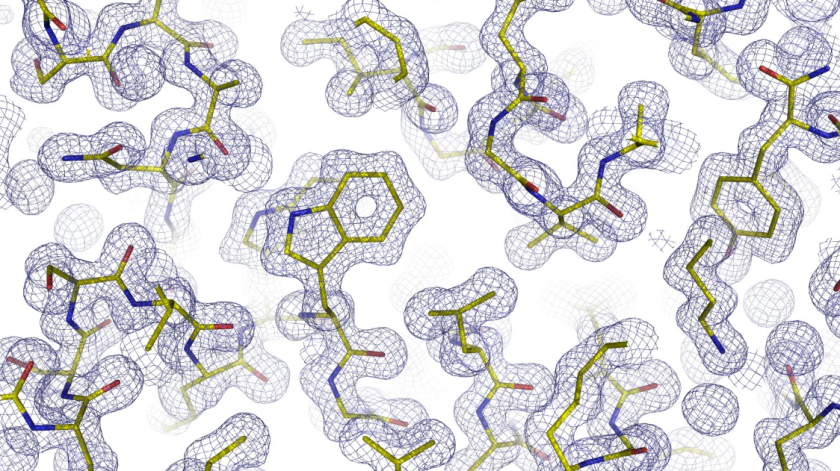
60s on the new detector were sufficient to obtain the electron density of the PETase enzyme. © HZB

The MX-beamline 14.1 has been upgraded with a new, better, faster and more sensitive PILATUS-detector.
© HZB
Last week a new detector was installed at one of the three MX beamlines at HZB. Compared to the old detector the new one is better, faster and more sensitive. It allows to acquire complete data sets of complex proteins within a very short time.
Proteins consist of thousands of building blocks that can form complex architectures with folded or entangled regions. However, their shape plays a decisive role in the function of the protein in the organism. Using macromolecular crystallography at BESSY II, it is possible to decipher the architecture of protein molecules. For this purpose, tiny protein crystals are irradiated with X-ray light from the synchrotron source BESSY II. From the obtained diffraction patterns, the morphology of the molecules can be calculated.
Now the MX team at BESSY II has put a new detector into operation at the MX beamline 14.1, which works about 2 to 3 times faster than before. The team analysed a crystal from the enzyme PETase as a sample. PETase does partially degrade the plastic PET. In less than a minute, the detector was able to record a complete diffraction data set, which includes data from an angular range of 180 degrees. The data set consists of 1200 images, each of which was exposed to X-rays for 45 milliseconds. "The resulting electron density was of excellent quality and showed all structural features of the enzyme," explains Dr. Manfred Weiss, who leads the MX team at BESSY II.
The success of the HZB MX beamlines is documented by more than 3000 PDB entries from experimental beamtime from more than a hundred international user groups from academia and pharmaceutical research companies.
red.
https://www.helmholtz-berlin.de/pubbin/news_seite?nid=21100;sprache=en
- Copy link
-
BESSY II: How intrinsic oxygen shortens the lifespan of solid-state batteries
Although solid-state batteries (SSBs) demonstrate high performance and are intrinsically safe, their capacity currently declines rapidly. A team from the TU Wien, Humboldt-University Berlin and HZB has now analysed a TiS₂|Li₃YCl₆ solid-state half-cell in operando at BESSY II using a special sample environment that allows for non-destructive investigation under real operating conditions. Data obtained by combination of soft and hard X-ray photoelectron spectroscopy (XPS and HAXPES) revealed a new degradation mechanism that had not previously been identified in solid-state batteries. They have gained some surprising insights, particularly regarding the harmful role played by intrinsic oxygen. This study provides valuable information for improving design and handling of such batteries.
-
Spintronics at BESSY II: Real-time analysis of magnetic bilayer systems
Spintronic devices enable data processing with significantly lower energy consumption. They are based on the interaction between ferromagnetic and antiferromagnetic layers. Now, a team from Freie Universität Berlin, HZB and Uppsala University has succeeded in tracking, for each layer separately, how the magnetic order changes after a short laser pulse has excited the system. They were also able to identify the main cause of the loss of antiferromagnetic order in the oxide layer: the excitation is transported from the hot electrons in the ferromagnetic metal to the spins in the antiferromagnet.
-
Environmental Chemistry at BESSY II: Radicals in waterways
How do radicals form in aqueous solutions when exposed to UV light? This question is important for health research and environmental protection, for example with regard to the overfertilisation of water bodies by intensive agriculture. A team at BESSY II has now developed a new method of investigating hydroxyl radicals in solution. By using a clever trick, the scientists gained surprising insights into the reaction pathway.
