Department Interface Design
Analytical methods
Laboratory-based characterization
Our laboratory at HZB is equipped with a multi-chamber ultra-high vacuum (UHV) system that features a glovebox, various manipulation and preparation chambers and two analysis chambers that are equipped with various laboratoy-based excitation sources allowing for sample (pre)characterization independent of available synchrotron beamtime. It allows for manipulating, treating, preparing, and analyzing samples without breaking the vacuum.
Photoelectron spectroscopy
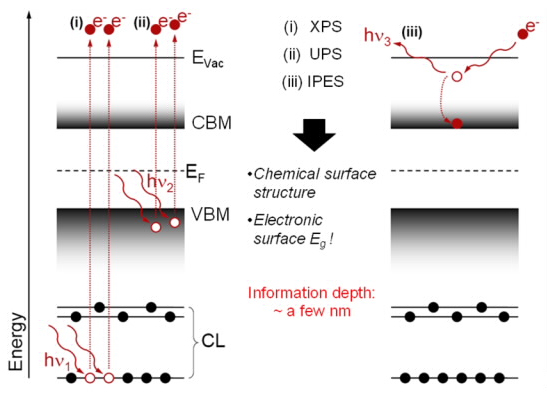
Photoelectron spectroscopy (PES) is an analytical tool for the characterization of the occupied electronic states of a sample. It is used in the analysis of elemental surface compositions and valence structures. In the PES process, one electron is excited from its ground state to a state above the vacuum level (EVac), where its kinetic energy (Ek) is measured by an electron analyzer with respect to the Fermi energy (EF). Depending on the source of excitation, PES is also called x-ray photoelectron spectroscopy (XPS) or ultraviolet photoelectron spectroscopy (UPS).
Inverse photoelectron spectroscopy
Inverse photoemission spectroscopy (IPES) is a method for probing the unoccupied electronic states of a sample. It is the inverse process to PES, i.e. electrons from above the vacuum level couple into unoccupied states of the conduction band, subsequently decaying into lower unoccupied states emitting UV photons, which are detected.
UPS and IPES are used to determine the position of the valence band maximum (VBM) and the conduction band minimum (CBM), respectively. Hence, by combination of these techniques it is possible to directly measure the electronic surface band gap of the sample.
Because of the short inelastic mean free path of the electrons, PES and IPES are very surface-sensitive techniques (information depth: a few nm).
Synchrotron-based characterization
In addition to our lab-based techniques (PES, IPES), we also use synchrotron-based characterization methods. The synchrotron-based characterization efforts are based on regular experimental campaigns at the synchrotron light source in Berlin (BESSY II) where we operate the HiKE endstation and the SISSY and OÆSE endstations at EMIL. In addition, we also perform experiments at the ALS, at ALBA, at Elettra, at Diamond, at Max IV, at SPring-8, and at the CLS.
X-ray emission spectroscopy
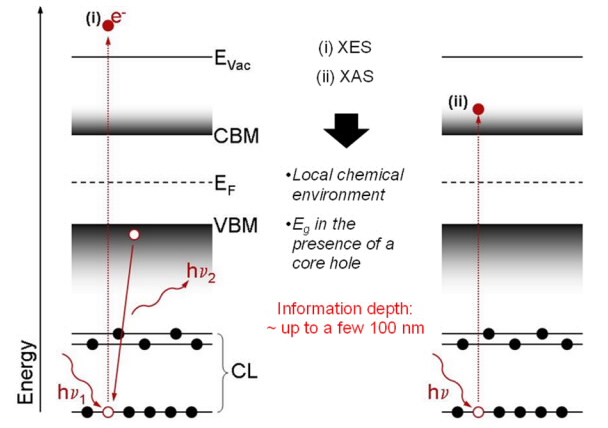
As PES, the x-ray emission spectroscopy (XES) probes occupied states and hence can be used to investigate the chemical and electronic structure of samples. For XES a core-level electron is excited by (soft) x-ray photons. The created core hole is filled by an outer-level (most often a valence) electron, causing the emission of a photon, which is detected by an x-ray spectrometer. The cross section for this process (compared to that of, e.g., direct photoemission) is very small, demanding the experiments to be performed at high-flux synchrotron light sources. Because the radiative relaxation process takes place with respect to the created core hole, XES is an excellent tool to probe the local structure of a specific atomic center.
Since XES (contrary to PES) is a photon-in/photon-out technique, the information depth is roughly two orders of magnitude larger than that of PES.
When the core-level electron is excited to the lowest unoccupied state of the samples conduction band, it is referred to as resonant inelastic x-ray scattering (RIXS). RIXS is used to map the electronic sample structure and is therefore uniquely suited to “test” calculated band structures.
X-ray absorption spectroscopy
X-ray absorption spectroscopy (XAS) investigates unoccupied states. It is an element-specific technique, sensitive to bonding environment and geometry. XAS involves the excitation of core-level electrons into unoccupied states of the sample's conduction band. For these measurements, the excitation energy is varied in distcrete intervals around the core absorption level of a specific element in the sample. The fraction of absorbed photons at each energy step is recorded through monitoring of secondary processes, resulting in different flavors of XAS (e.g. fluorsecence yield, FY-XAS or electron yield, EY-XAS). Depending on what detection channel is used, the XAS information depth is similar to that of XES (when using FY) or of PES (when using EY).
Similar to PES and IPES, also XES and XAS can be combined to reveal the band gap* of the near-surface bulk of the sample.
More information about the methods uses by the interface design department can be found at the EMIL webpage here.