Joint Research Group Macromolecular Crystallography
Structure of the month - February 2005
Nature Structural & Molecular Biology Vol. 12(1), 2005, Pages 90-96
Crystal structure of the polysialic acid degrading endosialidase of bacteriophage K1F
Katharina Stummeyer1, Achim Dickmanns2, Martina Mühlenhoff1, Rita Gerardy-Schahn1, and Ralf Ficner2
1 Abteilung Zelluläre Chemie, Medizinische Hochschule Hannover, Carl-Neuberg-Str. 1, 30625 Hannover, Germany
2 Abteilung für Molekulare Strukturbiologie, Institut für Mikrobiologie und Genetik, Georg-August-Universität Göttingen, Justus-von-Liebig-Weg 11, 37077 Göttingen, Germany
Abstract
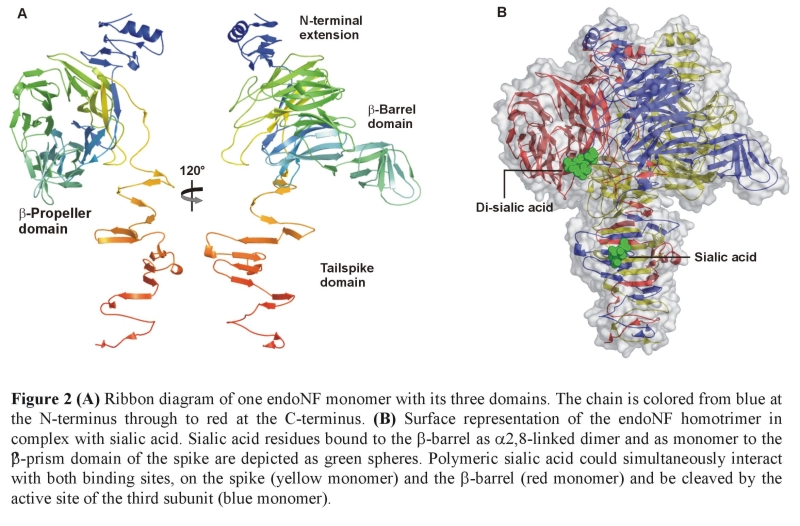
Phages infecting the polysialic acid (polySia) encapsulated human pathogen Escherichia coli K1 are equipped with capsule degrading tailspikes known as endosialidases, which are yet the only identified enzymes that specifically degrade polySia. As polySia is also an important promoter of cellular plasticity and tumour metastasis in vertebrates, endosialidases are widely applied in polySia related neurosciences and cancer research. Here we report the crystal structures of endosialidase NF and of its complex with oligomeric sialic acid. The structure, which reveals three distinct domains, indicates that the unique polySia-specificity evolved from a combination of structural elements characteristic of exosialidases and bacteriophage tailspike proteins. The endosialidase assembles into a catalytic trimer stabilized by a triple b-helix. Its active site differs markedly from that of exosialidases, indicating an endosialidase-specific substrate binding mode and catalytic mechanism. Residues essential for endosialidase activity were identified by structure-based mutational analysis.