Joint Research Group Macromolecular Crystallography
Structure of the month
Nature Vol. 471 (7340):651-52011 (2011)
Crystal structure of Metarhodopsin II
Hui-Woog Choe 1,2, Yong Ju Kim 1, Jung Hee Park 1,#, Takefumi Morizumi 1,#, Emil F. Pai 3, Norbert Krauß 4, Klaus Peter Hofmann 1, Patrick Scheerer 1 & Oliver Ernst 1,#
1 Institut für Medizinische Physik und Biophysik - CC2, Charité - Universitätsmedizin Berlin, Charitéplatz 1, D-10117, Germany.
2 Department of Chemistry, College of Natural Science, Chonbuk National University, 561-756 Chonju, South Korea.
3 Departments of Biochemistry, Molecular Genetics and Medical Biophysics, University of Toronto, 1 King´ s College Circle, Toronto, Ontario M5S 1A8, Canada.
4 Queen Mary University of London, School of Biological and Chemical Sciences, London.
# Present adress: Departments of Biochemistry and Molecular Genetics, University of Toronto, 1 King´ s College Circle, Toronto, Ontario M5S 1A8, Canada.
*email: Patrick.Scheerer@charite.de and Klaus_Peter.hofmann@charite.de
Abstract
G protein-coupled receptors (GPCRs) are ubiquitous signal transducers in cell membranes, as well as important drug targets. Interaction with extracellular activating ligands (agonists) turns the seven transmembrane helix (7TM) scaffold of a GPCR into a catalyst for GDP and GTP exchange in heterotrimeric Gαβγ proteins, which transmits the signal into the cell interior. The photoreceptor rhodopsin, a typical class A GPCR, couples to the G protein transducin and bears its ligand 11-cis-retinal covalently bound via a protonated Schiff base to the opsin apoprotein [1]. Absorption of a photon causes retinal cis/trans isomerization and generates the agonist all-trans-retinal in situ. After early photoproducts, the active G protein-binding intermediate metarhodopsin II (Meta II) is formed, in which the retinal Schiff base is still intact but deprotonated. Dissociation of the proton from the Schiff base breaks a major constraint in the protein and enables further activating steps, including an outward tilt of TM6 and formation of a large cytoplasmic crevice for uptake of the interacting C terminus of the Gα subunit [1]. Owing to Schiff base hydrolysis, Meta II is short-lived and notoriously difficult to crystallize.
We therefore soaked opsin crystals with all-trans-retinal to form Meta II, presuming that the crystal’s high concentration of opsin in an active conformation (Ops*) [2-4] may facilitate all-trans-retinal uptake and Schiff base formation. As a result we are presenting the 3.0 Å and 2.85 Å crystal structures (Figure 1a), respectively, of Meta II alone or in complex with an 11-amino-acid fragment derived from the C-terminal key interaction site of Gα (GαCT2) [5]. The X-ray diffraction data were collected at BESSY beamline 14-2. The structure was solved by molecular replacement.
GαCT2 binds in a large crevice at the cytoplasmic side, akin to the binding of a similar Gα-derived peptide to Ops* [3, 4]. In the Meta II structures, the electron density from the retinal ligand seamlessly continues into the Lys 296 side chain, reflecting proper formation of the Schiff base linkage (Figure 1b). The retinal is in a relaxed conformation and almost undistorted compared with pure crystalline all-trans-retinal. By comparison with early photoproducts we propose how retinal translocation and rotation induce the gross conformational changes characteristic for Meta II. The structures can now serve as models for the large GPCR family.
[1] Hofmann KP, Scheerer P, Hildebrand PW, Choe HW, Park JH, Heck M, Ernst OP. A G protein-coupled receptor at work: the rhodopsin model. Trends Biochem Sci., 34 (11):540-552 (2009)
[2] Park JH, Scheerer P, Hofmann KP, Choe HW, Ernst OP. Crystal structure of the ligand-free G-protein-coupled receptor opsin. Nature, 454 (7201):183-7 (2008)
[3] Scheerer P, Park JH, Hildebrand PW, Kim YJ, Krauss N, Choe HW, Hofmann KP, Ernst OP. Crystal structure of opsin in its G-protein-interacting conformation. Nature, 455 (7212):497-502 (2008)
[4] Scheerer P, Heck M, Goede A, Park JH, Choe HW, Ernst OP, Hofmann KP, Hildebrand PW. Structural and kinetic modeling of an activating helix switch in the rhodopsin-transducin interface. PNAS, 106 (26):10660-5 (2009)
[5] Choe HW, Kim YJ, Park JH, Morizumi T, Pai EF, Krauß N, Hofmann KP, Scheerer P, Ernst OP. Crystal structure of Metarhodopsin II. Nature, 471 (7340):651-52011 (2011)
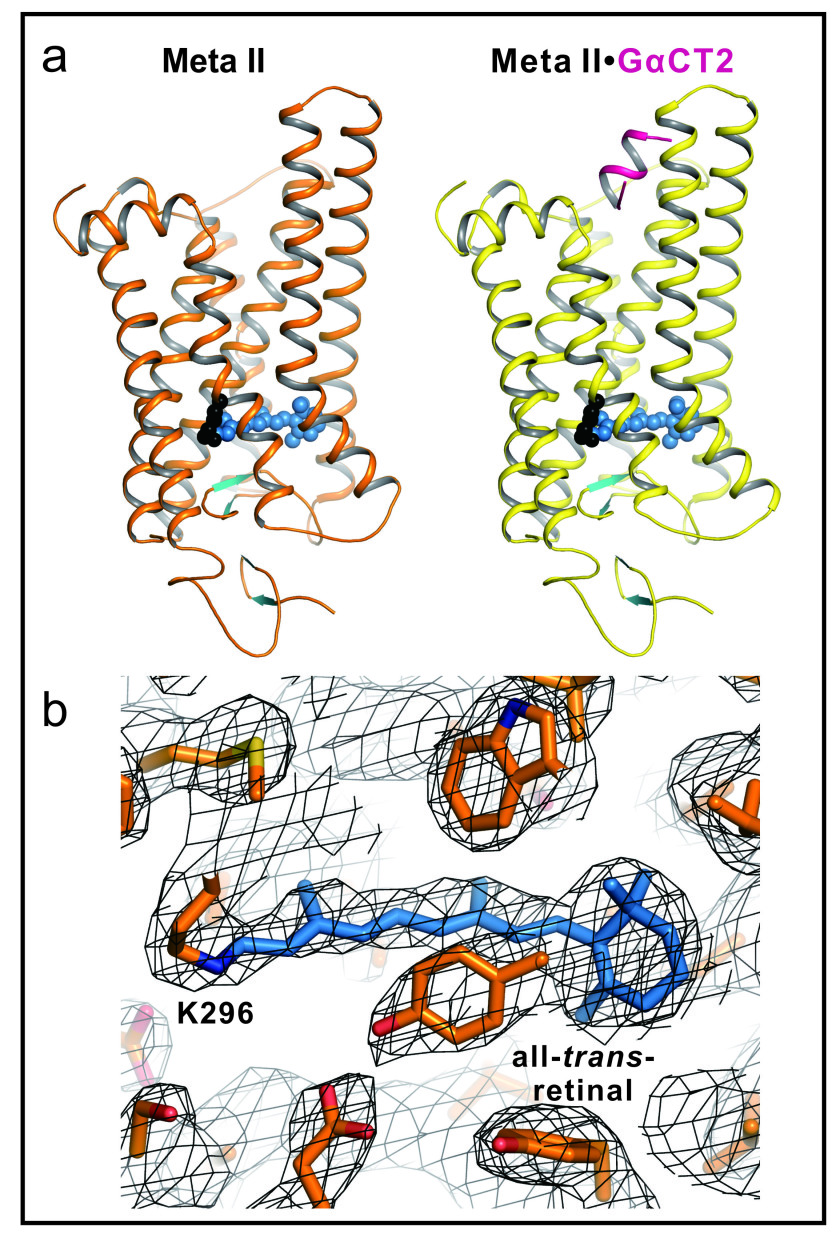
Figure 1.
(a) Active Rhodopsin structures of Meta II and Meta II in complex with a Gα fragment. Side view of cartoon model of Meta II and Meta IIGαCT2, that is, Meta II in complex with an 11-amino-acid C-terminal peptide derived from the transducin Gα subunit, representing the key binding site on the heterotrimeric G protein. In Meta II, the agonist all-trans-retinal (shown as blue spheres) is covalently linked to Lys 296. The G-protein-interacting conformation of Meta II (right panel) is identified by the bound Gα fragment (shown in purple).
(b) Retinal binding pocket of Meta II. All-trans-retinal and neighbouring residues shown as stick model for Meta II (orange) with 2Fo-Fc electron density map contoured at 1.0σ (grey mesh). Retinal is shown as blue stick model.