Joint Research Group Macromolecular Crystallography
Structure of the month - October 2004
Science Vol. 306, 2004, Pages 1390-1393
Anabaena Sensory Rhodopsin: A Photochromic Color Sensor at 2.0 Å
Lutz Vogeley1, Oleg A. Sineshchekov2,3, Vishwa D. Trivedi2, Jun Sasaki2, John L. Spudich2,4 and Hartmut Luecke1,5
1 Department of Molecular Biology and Biochemistry, University of California, Irvine CA 92697
2 Center for Membrane Biology, Department of Biochemistry and Molecular Biology, University of Texas Medical School, Houston TX 77030
3 Biology Department, Moscow State University, Moscow, Russia
4 Department of Microbiology and Molecular Biology, University of Texas Medical School, Houston TX 77030
5 Department of Physiology and Biophysics and Department of Informatics and Computer Sciences, University of California, Irvine CA 92697
Abstract
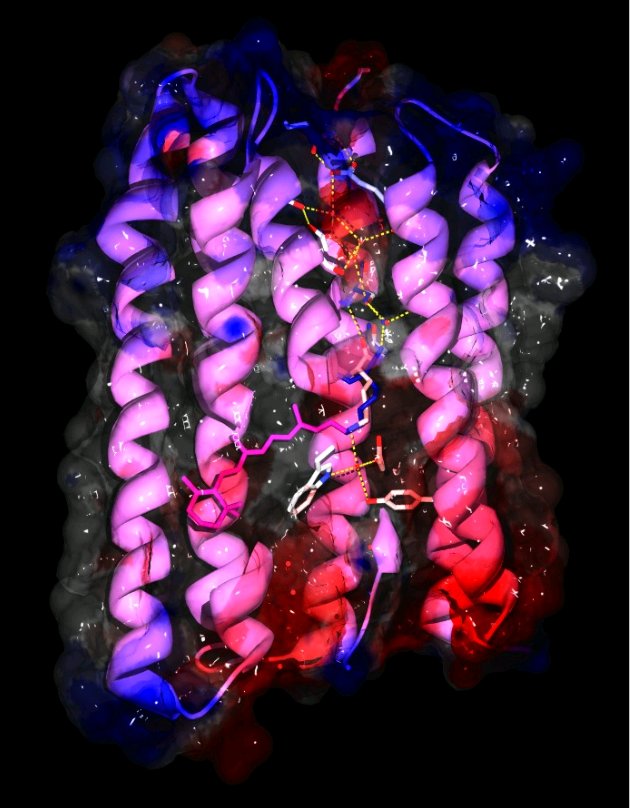
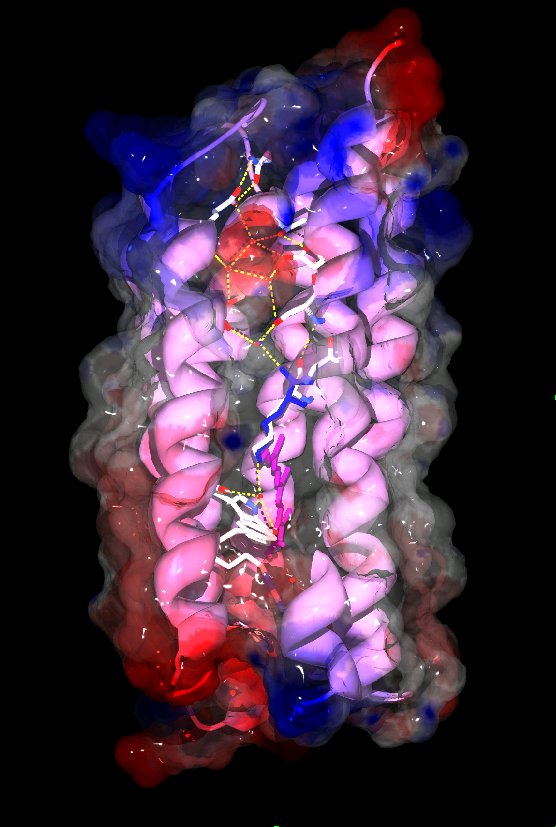
Microbial rhodopsins are membrane-embedded proteins with seven trans-membrane helices and a retinal chromophore covalently attached to a lysine via a Schiff-base.They have been found in archaea, eubacteria and single-celled eukaryotes and function as photo-sensors or light-driven ion transporters. The structures of several archaeal rhodopsins are available but as a cyanobacterial protein Anabaena sensory rhodopsin (ASR) is the first non-archaeal microbial rhodopsin to have its structure solved. It exhibits clear structural differences from the previously characterized archaeal rhodopsins, especially in the cytoplasmic half of the protein and the chromophore region, as well as functional differences. The retinal chromophore adopts stable 13-cis or all-trans states which can be interconverted by light. Since the all-trans to 13-cis ratio depends on the wavelength of the light used for illumination ASR could function as a color sensor and potentially regulate color-sensitive processes like the chromatic adaptation of photosynthetic pigment synthesis. Compared to the archaeal sensory rhodopsin II ASR features a much more hydrophilic cytoplasmic half with several embedded water molecules and polar residues that form an almost complete hydrogen bonded network from the chromophore to the cytoplasmic surface where ASR's soluble transducer is believed to interact. Alternate conformations of the carbonyl of the chromophore-bound lysine 210 in the electron density indicate a possible mechanism to complete the hydrogen-bonded network between chromophore and transducer binding site, which could be significant for transducer activiation.
The 13 lipid tails that could be built into the density at 2.0Å resolution indicate a significant distortion of the lipid bilayer from planarity in one dimension that has not been observed in cubic lipid phase crystals before.
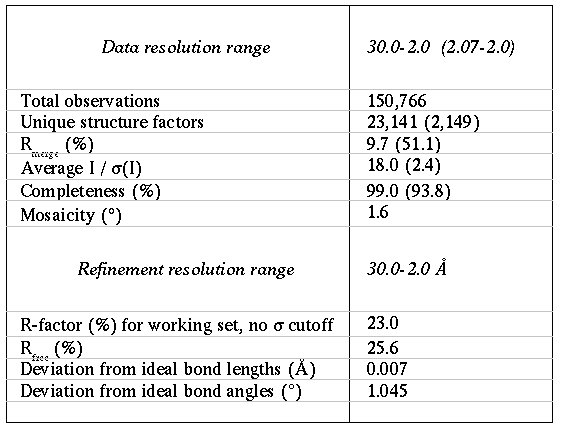
Data were collected at BESSY to 2.0Å and the structure was solved by molecular replacement using CNS and ARP/wARP at the beamline.