Joint Research Group Macromolecular Crystallography
Structure of the month - July 2004
Science Vol. 304, 2004, Pages 1672-1675
Phosphoryl transfer and calcium ion occlusion in the calcium pump
Thomas Lykke-Møller Sørensen1, Jesper Vuust Møller2 and Poul Nissen1
1 Department of Molecular Biology, University of Aarhus, Gustav Wieds Vej 10C, DK-8000 Aarhus C
2 Department of Biophysics, University of Aarhus, Ole Worms Allé 185, DK-8000 Aarhus C, Denmark
Abstract
The Ca2+-ATPase of the sarco(endo)plasmic reticulum (SR) is a primary transporter responsible for for the re-uptake of Ca2+ into the SR ultimately resulting in muscle relaxation. The cytoplasmic binding and luminal release of Ca2+ is fuelled by energy derived from ATP. During the enzyme cycle of the Ca2+-ATPase, it becomes phosphorylated (hence the denotion P-type ATPase). At least to distinct phosphorylated intermediates exist. A high energy phosphorylated intermediate (denoted E1P) which readily donates its phosphate back to ADP and a low energy intermediate (E2P) which does not. The conversion between the two phosphorylated intermediates is coupled to the translocation of two Ca2+ ions across the membrane barrier, i.e. the transformation of the cytoplasmic oriented high-affinity Ca2+ sites to luminal oriented low-affinity sites.
We have solved two structures of the Ca2+-bound Ca2+-ATPase at 2.6 and 2.9 Å resolution in complex with i) a nonhydrolyzable ATP analog (AMPPCP) and ii) ADP plus aluminium fluoride, showing how it reacts with ATP by an associative mechanism mediated by two Mg2+ ions to form an aspartyl-phosphorylated intermediate state (Ca2E1~P). The conformational changes that accompany the reaction with ATP pull the transmembrane helices 1 and 2 and close a cytosolic entrance for Ca2+, thereby preventing backflow before Ca2+ is released on the other side of the membrane.
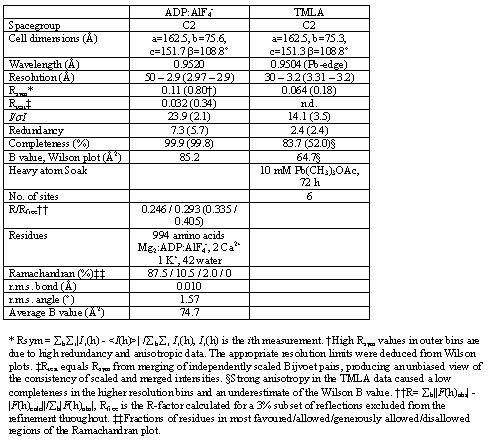
Diffraction was anisotropic, exhibiting 2.2 - 2.8 Å or 2.5 - 3.0 Å maximum resolution of stronger reflections in different directions of the AMPPCP and ADP:AlF4- forms, respectively. The AMPPCP form was used for experimental phasing using the trimethyl-lead acetate derivative (TMLA). SIRAS phasing based on heavy atom refinement against isomorphous differences at 30 - 3.2 Å resolution, and SAD phasing based on heavy atom refinement against anomalous differences at 30 - 3.8 Å resolution were combined and sharpened by a negative B-factor of 50 Å2 prior to density modification by solvent flipping and histogram matching at 2.6 Å resolution against native amplitudes as implemented in CNS. The experimental electron density map allowed for chain tracing of most parts. The refined AMPPCP form was used as the starting model for structure determination and refinement of the ADP:AlF4- form at 2.9 Å resolution, for which the same subset of reflections was used for calculation of Rfree.
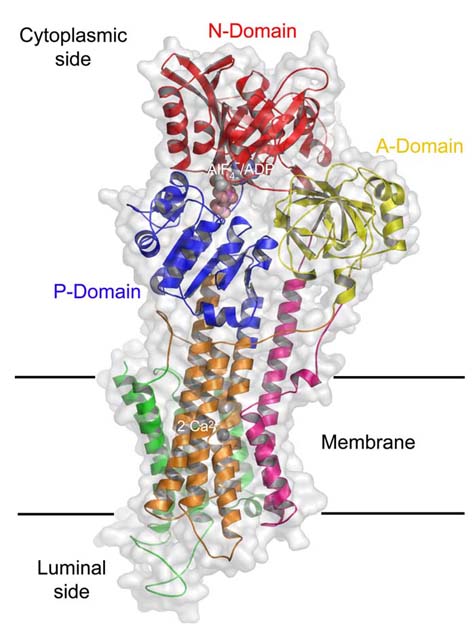
Structural cartoon showing the domain arrangement of the Ca2+-ATPase. Colours refer to individual domains with the A-domain in yellow, the M1-M2 domain in magenta, the M3-M6 domain in orange, the P-domain in blue, the N-domain in red, and the C-terminal M7-M10 domain in green.
If you have QuickTime installed, you can load a movie showing the conformational changes in the Ca2+-ATPase upon nucleotide binding. The transition is based on a simple linear interpolation between the Ca2-E1 and Ca2-E1-AMPPCP forms.